Advertisement
Tainted Weight-Loss Supplement Still Widely Used Despite Recall
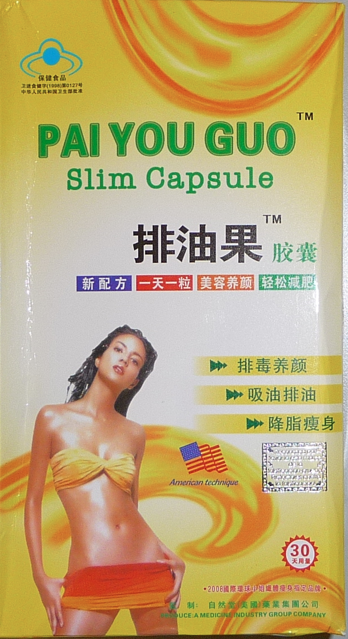
In the great battle between government regulators and dietary supplement peddlers, some think the regulators are losing.
According to Harvard Medical School researchers, a slew of dietary supplements contaminated with "a dangerous mix of banned pharmaceuticals" continue to be widely sold and used by women in the Boston area despite regulatory efforts to stop sales of the illegal products.
Indeed, nearly a quarter of the more than 550 women surveyed in the Harvard study were using or had previously used one product in particular, Pai You Guo, a weight-loss supplement made in China and known to be adulterated with the appetite suppressant sibutramine, which was pulled from the U.S. market because of increased risk of heart attack, stroke and cancer, and the laxative phenolphthalein, withdrawn from over-the-counter sales in 1997 "due to its carcinogenic properties," the study says.
The U.S. Food and Drug Administration announced a "voluntary" recall of Pai You Guo in 2009, but researchers say the move had "little effect" on the number of women using it.
Study author Pieter Cohen, a general internist at Cambridge Health Alliance and Assistant Professor of Medicine at Harvard Medical School said that despite intense outreach by doctors and the FDA about the perils of taking such contaminated products, not a single woman interviewed in the study was aware of the FDA action. Moreover, the majority of those surveyed began taking Pai You Guo after the recall went into effect, he said.
Participants were a group of Brazilian-born women who attended a primary care clinic in Somerville, Mass., or one of several nearby churches. (Cohen said there was evidence that the weight-loss supplements were popular among these immigrants.) Researchers found that participants who were overweight, obese or lacked health insurance were more likely than normal weight and insured women to report taking Pai You Guo, which means "the fruit that sheds the fat" in Mandarin.
The study, published in the Journal of General Internal Medicine, focused only on Pai You Guo, a poster child for dangerous, drug-laden, over-the-counter products masquerading as natural slimming aids. (With nice marketing too, after all, who wouldn't want to look like the hot, svelte, bikini-clad model on the box?) Taken as a capsule, or as a tea, Pai You Guo has been featured in news stories, and the general problem of contaminated supplements has been widely reported, for instance, in The New England Journal of Medicine, among other publications. But Cohen says there are literally hundreds of different tainted weight-loss supplements, with names like Super Slim and Planta Fruita, sold in shops around town.
"If you walk into Allston today, you'll see two dozen products that look suspicious," he said.
Cohen said his study, which sought to verify previously anecdotal evidence about continued sales of the product, revealed several surprises.
First, he said, he was shocked to see that retail, rather than Internet sales were driving consumers, and that the majority of Pai You Guo sales were from neighborhood mom-and-pop shops or through individual distributors.
He also said he was surprised and concerned about how ineffective the FDA's actions have been at putting a dent in sales.
"The regulatory framework just isn't working," Cohen said, "and we're going to have to reform how we regulate supplements in order to make sure they're safe. Right now, anyone can sell anything, as long as they name it a 'dietary supplement,' and the FDA has to play catch up. That's just daunting."
To be fair, trying to shut down the illicit dietary supplement trade is a little like attacking the many-headed hydra of Greek mythology: every time you cut off one head, two more grow back.
FDA spokesperson Siobhan DeLancey said for one thing, tracking down the actual manufacturer of the products requires painstaking sleuthing: the packaging is often not in English and the manufacturer's name is often not listed. So, investigators must start at the bottom of the distribution chain and slowly work their way back to the source. "And there can be hundreds of distributors," DeLancey said.
But the main problem with supplements, she said, is that they're not regulated like pharmaceuticals. Under current U.S. law, supplements can be sold on the market without being reviewed for safety and effectiveness, as is required for drugs. "People exploit that loophole to put out products that aren't really dietary supplements," DeLancey said.
For instance, even if the FDA is able to crack down on one product, like it did in the case of a supplement called "2-Day-Diet," another manufacturer will start making it in a slightly different way. "If a product has an established following, as soon as the first person gets busted other people bounce up," DeLancey said. "They take the names, take the packaging and make something similar to capitalize on it."
Indeed, negative side effects don't seem to slow demand.
The Harvard researchers report that the great majority of women using Pai You Guo (85%) experienced at least one side effect, including dry mouth, insomnia, palpitations, fainting, anxiety or addiction to the product. But, according to Cohen, 56% of the woman also said they lost weight.
"The great majority of women who take it believe it's safer than a prescription to help them lose weight," Cohen said. "They think because they can buy it over the counter, it's safe."
In his 2009 New England Journal piece, Cohen explains how supplement makers are devising clever new ways to elude regulators:
Recently, unscrupulous manufacturers have made it more difficult for the FDA to detect undeclared ingredients by incorporating pharmaceutical analogues into their products. Analogues are created by modifying the original chemical structure of a compound — for example, by adding a hydroxyl group. It is suspected that these analogues are developed to evade detection by the FDA, making the products more difficult to regulate, and to reduce the risk of patent-infringement lawsuits. A recent analysis showed that more than half of 26 supplements marketed for the enhancement of sexual function contained analogues of phosphodiesterase type 5 inhibitors. Because these analogues have never been studied in humans, their risks are unknown, but unexpected adverse reactions to analogues have already been documented. Reports from Britain, China, and Japan, for instance, link an analogue of fenfluramine to liver damage, including fulminant hepatic failure necessitating transplantation.
"The bottom line," he says, "is that these products are dangerous and shouldn't be used."
As for the regulators, they've decided that in addition to going after the supplement sellers, they ought to try harder to reach consumers. So, here's the FDA's trove of information on tainted dietary supplements; and here's a link to sign up for a personal email message every time the agency issues an alert on a tainted supplement.
This program aired on August 16, 2011. The audio for this program is not available.
