Advertisement
Waltham Company's Lobbying Effort For Its Drug Used To Treat Opioid Addiction Raises Questions
Resume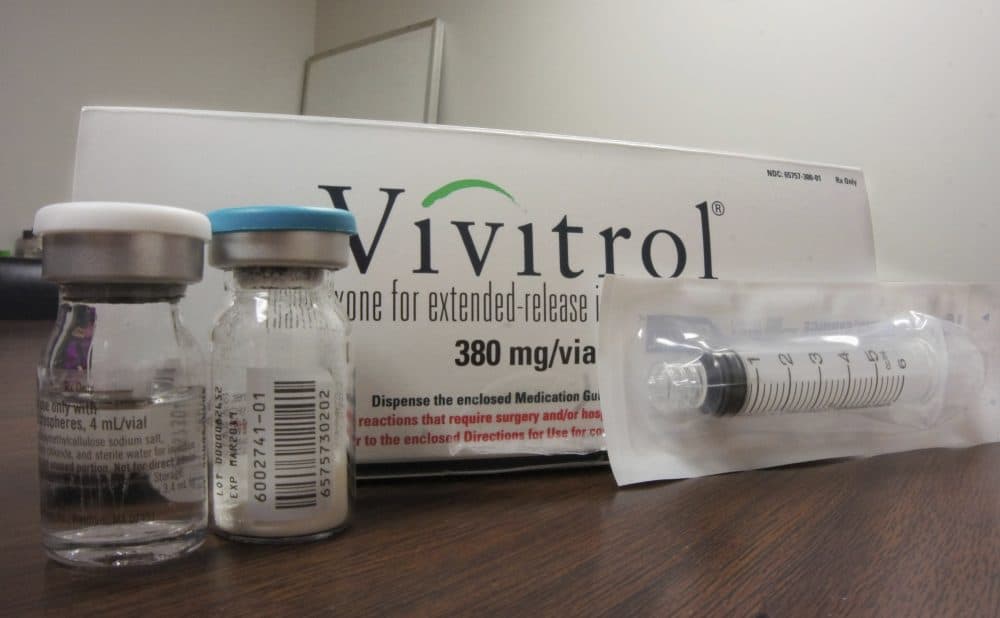
A NPR investigation published Monday raises questions about the marketing and political influence surrounding a drug used to treat opioid addiction.
The drug, produced by the Waltham-based company Alkermes, is designed to block the brain's receptors from being able to feel an opioid high. It's known by the brand name Vivitrol and the generic name naltrexone.
NPR's investigation found Alkermes lobbied in several states for legislation promoting Vivitrol, while also pushing for stricter regulations on competing addiction treatments.
Massachusetts state records show the company and its CEO, Richard Pops, made dozens of donations through its political action committee to Massachusetts political and law enforcement leaders. (Details here and here.) One is Middlesex County Sheriff Peter Koutoujian, who spoke about Vivitrol — and why he thinks it's better than a different treatment, buprenorphine, also known by the brand name Suboxone — to a group of Alkermes investors in September.
"Some of our security issues with buprenorphine is that it is our most smuggled drug," Koutoujian said at the time. "It is atomized. It is soaked into papers. It is put under tabs. It used to be put under stamps. This is a smuggled drug — a smuggled piece of contraband that has a black market value inside and uses for which it's not intended. I wanna make a note that no one is smuggling naltrexone or Vivitrol. It has no value inside or outside."
Koutoujian stands by his decision to use Vivitrol in his jails. His office says 80 percent of all participants who volunteered to take part in a six-month post-release program involving Vivitrol have not relapsed.
Aside from its use in jails and at some state addiction treatment centers, legislation was filed on Beacon Hill this year involving the drug.
The bill, sponsored by Rep. Elizabeth Malia, who represents parts of Boston, would expand training for medical professionals to administer Vivitrol.
Records show that Alkermes' PAC donated $1,000 in total to Malia's campaign.
A request for comment to Malia's office on Tuesday was not returned.
That's disturbing to some addiction specialists who say Vivitrol is a new drug and hasn't been studied well yet — particularly its efficacy compared to other treatments like Suboxone and methadone.
"I think it's important to counter an inflated notion of efficacy of this medicine," said Dr. Michael Bierer, a physician at Massachusetts General Hospital and president of the Massachusetts chapter of the American Association of Addiction Medicine. "It works and for some people it works beautifully, and it's hard to predict for whom it will be a great drug, but people in the latest study that I know of who were treated without naltrexone had a remission rate of 40 percent versus people without naltrexone at 60 percent."
In a statement to WBUR, Alkermes said opioid addiction is a "complex disease" with no single, correct way to treat it.
"Ultimately, it is critical that patients and families should have access to all FDA-approved medicines for opioid addiction, and that treatment providers should know the alternatives and make the most appropriate treatments available without bias," the statement said.
Pam Wilmot, the executive director of Common Cause Massachusetts, a nonprofit focused on government accountability, joined WBUR's Morning Edition to discuss Alkermes' lobbying efforts. Listen to that interview at the top of this post.
This article was originally published on June 13, 2017.
This segment aired on June 13, 2017.

