Advertisement
Q&A: Putting The Shingles Vaccine On Your Medical To-Do List At 50
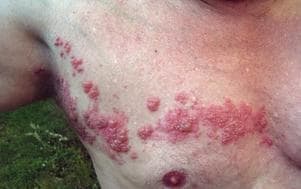
I’m terrified of shingles. My grandmother described the pain as the most excruciating of her life, far worse than childbirth. A family friend not only had shingles but a hideous after-effect called post-herpetic neuralgia, which I would translate as "endless torture," or perhaps "hell on earth" — months and months of unbearable agony. (Read the basics on shingles here.)
So when the news broke late last week that the FDA had lowered the cut-off for shingles vaccine approval to age 50 and above — the previous cut-off had been 60 — I wondered how quickly the new federal guidelines would translate into actual vaccines at our doctors' offices. Since it was first approved in 2006, the shingles vaccine — called Zostavax and made by Merck — has brought a mix of headlines: some laments that seniors were not taking full advantage of it, other complaints that shortages made the supplies spotty, and that insurance coverage was spotty as well.

Q: Why has the recommended cut-off just dropped from age 60 to age 50?
A: Clinical trials have now demonstrated that the vaccine's benefits extend down to age 50, so that has changed the recommendations of the CDC vaccine panel. It's not that anyone thought it wouldn't be beneficial to younger people, but companies seeking licensure try to pick a group in which there's a very clear and dramatic effect, and it's very clear that shingles is much more common in older people. All along there has been the thought that they probably would push it younger, and that's where we are now. There will undoubtedly be some age at which it's not worth doing, but at this point the cut-off is 50.
Q: How are you translating this for your patients? Will you move immediately to suggesting the vaccine for patients between 50 and 60?
A: Before we make a widespread recommendation, we want to wait and be able to tell our patients whether health insurers will pay for it or not. It's likely they will. In general, the health insurers follow the CDC vaccine recommendations, though with expensive vaccines like this — Zostavax costs about $200 — there are starting to be some holes in that patchwork.
Q: Do they pay for it now for 60 and above?
A: Most do. All the major payers that we work with do cover it. The complication comes for Medicare patients: It's not that it's not covered, but it's covered under Medicare Part D, which makes the payment for the vaccines very complicated instead of being straightforward. The patient has to take the prescription for the vaccine from the doctor to the pharmacy, and then bring the vaccine — which has to be kept frozen — back to the office to have it administered.
Q: That sounds stupid. Is there a movement to change that?
A: As far as I know, there isn’t. But I've heard of at least one company springing up that is trying to handle all the paperwork and make it possible for doctors' offices to just stock the vaccine and get reimbursed for it.
Q: So what should fiftyish people do at this point?
A: Very few people are likely to be at imminent high risk of developing shingles. I’d recommend people wait a few weeks for the insurance companies to make up their minds, then call the company to make sure it's covered.
Q: Once they are sure it's covered, is it likely to be available? There have been reports of Zostavax shortages.
A: That's still happening. The vaccine uses extraordinarily large quantities of the attenuated virus and it's hard for the company to keep up with demand, and it has been since they first created the vaccine. I think the manufacturer is now shipping orders placed about three or four months ago.
Q: How does the vaccine work?
A: This is a typical attenuated live viral vaccine similar to the chicken pox and measles vaccine. So it introduces a weakened version of the virus into the body, and that virus reproduces briefly and stimulates the immune system to recognize it and "put out the fire" of the attenutated virus, and strengthens the immune response against the reactivation of the virus in the future. Shingles is a reactivation of the chicken pox virus. Most commonly, when you recover from chicken pox, the virus goes into hiding, becomes dormant and lives in a suppressed state in the body. At some point in the future, it can reactivate, and what commonly reactivates it involves illness, stress, drugs that suppress the immune system or just the waning of the immune response with age.
What the vaccine does is essentially gives your immune system the proverbial shot in the arm.
Q: How would you sketch out the risk-benefit profile of Zostavax to a patient?
A: There really is very little risk with this vaccine. The only situation where I'd have concern about it would be somebody who was profoundly immuno-suppressed by some major illness or potent medication.
Q: And the kind of post-herpetic neuralgia our family friend had is a sort of worst-case scenario of shingles?
A: Yes. The average person who gets shingles is quite uncomfortable, but it's fairly brief. There's really a huge range. Post-herpetic neuralgia is probably the strongest reason for taking this vaccine: it's not super-common but it certainly happens enough to justify the use of the vaccine. and it is a persistent and excruciatingly painful condition where, for reasons we don't really understand, the reactivation of the virus triggers a really hyperactive pain response. It can lasts months, many months, even. There are treatments, but they’re not a sort of simple, straightforward, take-this-pill-and-you’re-cured type of treatment.
Readers, is there anything I forgot to ask? Please post questions in the comments below. Basic FDA information about Zostavax is here. And more background, including a vivid description of shingles, is here.
This program aired on March 28, 2011. The audio for this program is not available.
