Support WBUR
Former Regulator Calls For More Oversight Of Compounding Pharmacies
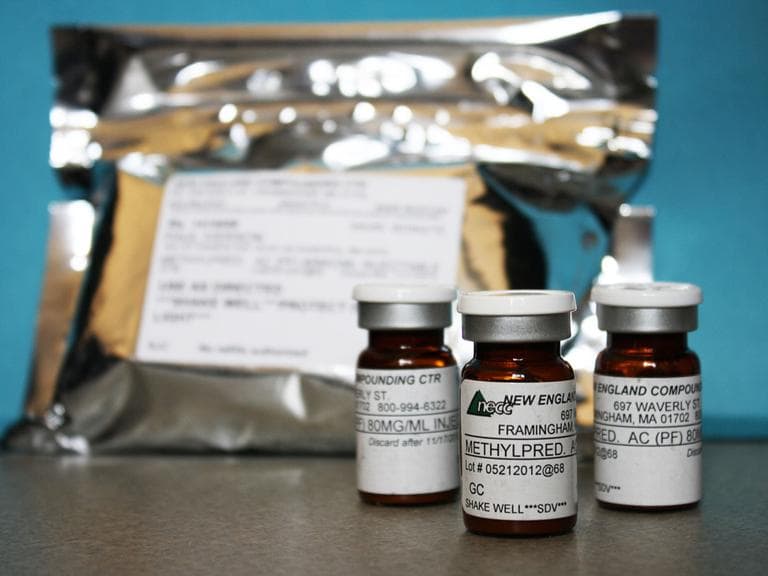
Federal health officials are now urging doctors to warn all patients who have received any injectable drug made by the Framingham compounding pharmacy tied to a nationwide outbreak of fungal meningitis. Fifteen people have died and 214 people have become ill in 15 states.
The FDA says three new cases of infection could have been caused by products other than the suspected steroid.
The outbreak has raised questions about the little known sector of "compounding pharmacies," which take medications and personalize them for individual patients. For a look at the industry's regulations and oversight, WBUR's Morning Edition host Bob Oakes spoke with a former member of the Massachusetts Board of Pharmacy, Bill Gouveia. He currently teaches at the Northeastern University School of Pharmacy.
Bill Gouveia: We know how to do this job very well: namely to prepare the proper drug in the proper way in a sterile manner. We know how to do that. That's in the pharmacy literature goes back 20, 30 years. The problem is we haven't taken knowledge and said to the consumer, "This is what you should look for when you look at a pharmacy," and to the state regulators to say, "This is what you should adopt in terms of the standard of quality."
Bob Oakes: But if a compounding pharmacy is licensed to operate in Massachusetts, why wouldn't the consumer just assume that it's complying with all the regulations, that it is a sterile and safe place, that it's producing safe drugs?
I think the consumer needs to be vigilant. Much as they were with medication errors and prescription misfills, they now are more aware that they should understand the drug and how it's used and who dispensed it and who made it.
The federal Food and Drug Administration [FDA] says there needs to be clearer regulations, that industry basically grew beyond what they can currently regulate. Do you agree with that?
Yes. There's disclarity about what is within the state's purview and what's in the federal purview. If you're compounding in Massachusetts alone, then your regulatory authority is the [state] Board of Pharmacy. Once those drugs go out of Massachusetts, then the FDA is responsible for that evaluation. And I'm not sure they have resources to do this properly.
What about the state. Does the state have the resources?
No, they really don't. The state, for example, the Board of Pharmacy — who works hard to do their best — has to inspect 1,100 pharmacies in the commonwealth. And really they can only respond to specific complaints, so their response is really knee-jerk to what's out there in the field. But they don't have any systematic way, they don't enough resources to really look at these operations.
Are the state and federal governments, the regulators, placing too much trust on these businesses to regulate themselves?
Oh, I think that's the case. This operation just ran amuck for years dispensing thousands of prescriptions and no one really came in to say, "What are the safety requirements, what are the accreditation requirements that you're meeting?"
Well, that being the case, shouldn't there then be someone overseeing this actively and who should that be?
It probably should be the Board of Pharmacy...
On the state level.
On the state level and the FDA on the federal level. But, Bob, we still don't know what went on in Framingham. I think until we do a detailed analysis and really understand that these are the factors which caused the problem. We need to regulate around those problem areas, but we still don't know what went on.
Exactly what went on in terms of how the injectable steroids became contaminated, you mean.
Right. There needs to be presence and there needs to be the state authority that says to the pharmacists, "How are you meeting the high quality standards that we need?"
What do you think should be done about this?
I think we need to look at the resources required for the Board of Pharmacy, number one. Look at their regulations, number two. And, number three, establish a clear commitment on the commonwealth of being sure our patients are protected from these shoddy operators.
So does that mean the state Board of Pharmacy needs to get bigger and in some ways bolder in terms of how it approaches these companies?
Absolutely. Absolutely. Clearly, problems are addressed best before they occur. And so if you look at the regulations which are available, their application and evaluation on the part of individual pharmacies by the board is just not there.
So it's likely that they would not have found the fungal contamination of these injectable steroids before they were distributed anyway?
Without spending an awful lot of time in them. To some degree, Bob, it should be a case where the board goes unannounced into pharmacies to say, "Show me your regulations. Show me your training. Show me your facilities." They don't have the time and effort to do that.
Do you think the political will exists up on Beacon Hill to update state regulations and make them tougher and put more money into the state board of pharmacy at least in terms of the number of inspectors out there looking into these businesses?
I think so. The severity of what went on tells me that we really need to respond with all of our healthcare authority that we can bring to bear on this particular issue.
A spokeswoman for the Massachusetts Department of Public Health says the agency investigated more than 300 complaints last year, and reserves the right to inspect pharmacies at any time without notice.
This program aired on October 16, 2012.

