Support WBUR
Is Amyloid, The Leading Hypothesis For Treating Alzheimer’s, Played Out? Not So Fast
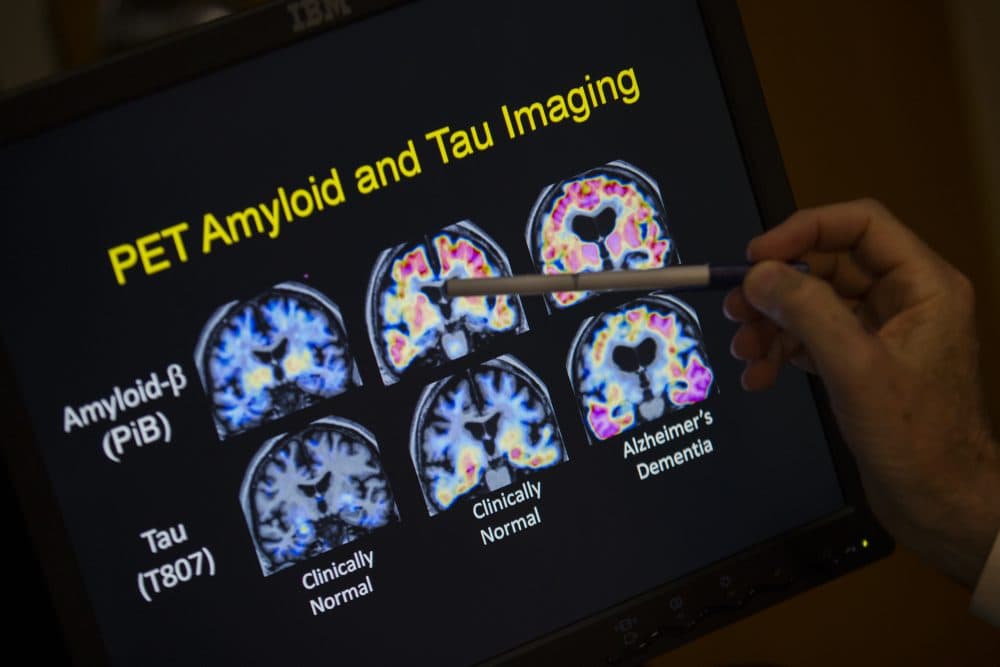
For decades, Alzheimer’s researchers have pursued the idea that clearing out clumps of a protein called amyloid would delay or cure the brain disease.
But in hundred-million-dollar trial after hundred-million-dollar trial, the approach hasn’t panned out.
In an editorial last week in the New England Journal of Medicine, Alzheimer’s researcher M. Paul Murphy of the University of Kentucky declared the impending death of this so-called amyloid hypothesis.
The failure of the latest trial of Eli Lilly’s anti-amyloid drug solanezumab was also reported in detail in the journal, though its negative results had been known for months.
“It would be foolish to ignore the continued failures of anti-amyloid approaches,” Murphy wrote in his editorial. “The field is clearly in need of innovative ideas. We may very well be nearing the end of the amyloid-hypothesis rope, at which point one or two more failures will cause us to loosen our grip and let go.”
But others in the field disagree that the amyloid hypothesis has been fully played out. There is still room for optimism, they said this week, that getting rid of amyloid can be the right treatment -- if given to the right patients at the right time.
Three large clinical trials have officially failed, but they all showed a hint of effectiveness, said Brigham and Women's Hospital neurologist Reisa Sperling, who co-directs the Harvard Aging Brain Study at Massachusetts General Hospital.
Sperling is capitalizing on that hint in a trial she’s running, giving solanezumab to more than 1,100 people who have some amyloid in their brains but who don’t have any of the devastating symptoms of Alzheimer’s, such as losing the ability to care for themselves along with decades of memories.
She spoke with WBUR about the amyloid hypothesis, her hopes for this trial and the future of Alzheimer’s treatment.
Do you agree that this study should tell us we’re near the end of the rope with the idea that treating amyloid can effectively treat Alzheimer’s?
I’ve been hearing about the death of the amyloid hypothesis for 20 years.
Why have all these studies failed to show a benefit from clearing amyloid out of the brain?
Unfortunately, this is one in a series of studies that suggests when you’re going after this amyloid plaque buildup, that you need to start 10 years before.
So these studies did too little, too late?
We’ve been doing trials like giving someone a statin when they’re in the cardiac ICU and they have heart failure. If you only tried statins in people who already have very damaged hearts, we would never have prevented all the heart attacks we have. So I feel like I’m not going to give up until we’ve tested it at the right stage of disease.
You’re running a trial now, called the A4 trial, for Anti-Amyloid Treatment in Asymptomatic Alzheimer’s. You’re going to be giving solanezumab to volunteers, ages 65-85, who have amyloid in their brains but haven’t yet developed symptoms of Alzheimer’s. Is that going to be early enough in the disease process?
That’s the thing that keeps me up at night, although I’m so excited about A4.
Should there be trials of people who are at even earlier stages, decades before symptoms appear?
The next trial I start — which is called the A3 study — I hope it’s going to start as young as age 50. Very few people at age 50 will have evidence of early amyloid buildup, but the idea is we’ll catch people who we think are just beginning to accumulate amyloid. They’re not positive on a PET scan yet, but because of their genetics and age and other things we learn about them, we think they’re at high risk. At that stage, we will start them on a drug to try to prevent them from accumulating amyloid to begin with.
But you’re still going forward with the A4 trial in people with signs of amyloid?
Even if A4 is not optimal, even if we should go earlier than that, if we slowed decline by 15 percent, we’d still prevent dementia in quite a number of people -- even if the train is already on the track.
Because Alzheimer's is a disease of old age, delaying it means someone will more likely die of something else first?
I always like to say they will die out ballroom dancing instead of in nursing homes, which is of course what they want and certainly from a public health point of view, what we need.
You’ve adjusted the A4 trial after seeing the results of this latest failure, right?
We quadrupled the dose [of solanezumab], which is a gutsy thing to do in the middle of a trial.
Secondly, we extended the trial because we wanted everybody to have a chance to have the dose escalation. We thought it was fair that people who’ve been in the study for two to three years already should have a chance to have the higher dose. [And] following people longer buys us the opportunity to see if we’re really slowing cognitive decline. We don’t want to be left with another study: Was it too little or too late?
So when will the trial be completed now?
2022. That’s hard. We still have something like four years, five months and three days until we have answers. But who’s counting?
Why has Alzheimer's proven so difficult? Why don’t we have a treatment or way to delay it, never mind a cure?
Alzheimer’s is a very complex disorder. Part of it is just that it’s a slowly, inevitably progressive disorder, and we only know parts of what might be involved. Secondly,
we’ve been looking at Alzheimer's at the end stage of the disease rather than looking at the beginnings and the risk factors. That’s what I hope the A4 study and other prevention studies are doing now.
Is money part of the problem?
We haven’t thrown enough resources at Alzheimer's disease. Until recently, we were spending one-tenth on Alzheimer's what we spend on cancer.
But Congress came through with extra money last year?
For the first time in a very long time, there was an increase in Alzheimer's funding, about $400 million. I saw this tremendous increase in exciting science.
Will one drug ever be enough to effectively treat Alzheimer’s, or should we be looking at combinations?
I really think we have to start combination therapies right now. There are therapies coming along that are going after the tau tangles [another hallmark of Alzheimer’s in the brain]. We could fail on both amyloid and tau because we’re not using them together. I’m very keen to start those combination trials.
What about lifestyle factors? Can we exercise, eat and sleep our way to a lower risk for Alzheimer’s?
The strongest data is on exercise. People staying physically, mentally, socially active really can help the resilience of your brain. It’s unlikely for Alzheimer’s that lifestyle and diet and exercise are going to be enough, but everyone should do it anyway, particularly for those people who have [close relatives with the disease].
PET scans of the brain can now see amyloid and therefore diagnose Alzheimer’s. If we still don’t have a treatment, why does that matter?
We now have the ability to see the changes in the brain years before symptoms. From my point of view that’s a glass half full. We can try to intervene during that 10-year silent period before people get to the terrible stage of Alzheimer’s disease that’s so devastating to their families.
What about the idea of a blood test for Alzheimer’s, which would be far less expensive than a PET scan. There have been some research advances but we’re still not there yet. Do you think we’ll get one soon?
I am convinced that over the next few years, we’ll have a blood test that substantially improves our ability to detect amyloid before people have symptoms, so I’m excited about that.
Lifestyle factors can defer but not prevent the disease, and there are no viable treatments yet, so is there anything people can do now other than wait?
One of the things people can do is come and volunteer for a clinical trial. I believe that the cure or at least an effective treatment is likely in someone’s test tube now, and the bottleneck is getting people into trials. Especially for people who might have a family history but they’re doing OK now — they can really help us.
How do you protect your own brain?
I try to go to dance class two to three times a week. I try to stay engaged and feel like my life has a purpose. I hope that some of the work we’re doing in Alzheimer’s will benefit me in time, because I have a very strong family risk, but mostly I hope it’s going to protect my daughters.
Even though some drug companies have pulled out of the search for a treatment, you’re going to continue fighting?
We can’t give up. What's the option?
