Support WBUR
Losing To Lyme
Our Dogs Can Get Lyme Vaccines And We (Still) Can't. When Might We?
Part of our Losing to Lyme series
For Dr. Stanley Plotkin, a prominent vaccine scientist, Lyme disease is personal. His son, Alec, collapsed from a slow heart rate when he was 39, brought down by a rare heart complication from Lyme.
His son survived, but the incident helped cement Plotkin's resolve to pursue a human vaccine against Lyme disease. Using his bully pulpit as an emeritus professor of infectious diseases at the University of Pennsylvania, he’s taken his case from The New York Times to the New England Journal of Medicine, in which he called the lack of Lyme protection "the worst recent failure to use an effective vaccine."
That’s because we used to have a vaccine for Lyme, called LYMErix, but it was pulled from the market. Now, the only family member who can get a Lyme vaccine is your dog.
LYMErix had some problems. It required three doses at $50 each, and they were not covered by insurance -- so involved some inconvenience and out-of-pocket money. Despite a good safety record in clinical trials, some people experienced what they thought were side effects and sued SmithKline Beecham, the manufacturer. In 2002, SmithKline pulled the vaccine, after only four years on the market. (More on the history of the Lyme vaccine here.)
While the official line is that poor sales led the vaccine's maker to pull it, most experts think the specter of lawsuits was a key factor. Though an FDA panel ultimately found no link between the vaccine and arthritis, SmithKline settled lawsuits making that claim. And by then, the vaccine was already dead.
That cautionary tale still reverberates at companies developing new potential Lyme vaccines. "When I talk to manufacturers, they essentially ask me: 'Will it happen again?' " Plotkin said.
Bring Back The Old Vaccine?
A number of scientists hope to change that. One is Tufts University professor Sam Telford, an expert on tick-borne diseases, who in the early 1990s organized one of the LYMErix clinical trials.
Telford hopes to bring back that same vaccine technology, knowing that pharmaceutical companies will be hesitant. “Pharma won’t touch LYMErix again,” he said. “It’ll take some quixotic wacko like me, under a nonprofit, to bring it back.”
“Why pay $150 to $200 million to bring a new vaccine to market?” Telford asked, likely even underestimating the typical vaccine R&D costs of up to $500 million a new Lyme vaccine could require. “For $10 million, the old vaccine could be pushed forward.”
Given his prior research, Telford knows the vaccine recipe. The patent rights have expired, so it would be relatively cheap to produce, he thinks.
But Plotkin thinks the safety concerns make the possibility of bringing the old vaccine back a non-starter. Instead, it’s better to start over, and eliminate the controversial parts of the old vaccine blamed for the alleged bad side effects, “even if the scientific evidence doesn’t justify it,” he said.
In 2014, the health care giant Baxter was working on a promising vaccine that fit Plotkin’s mold. It included vaccine targets that would work on both North American and European strains of Lyme, allowing them to expand the market to make the investment financially viable.
But Baxter decided instead to sell off its entire vaccines portfolio. A couple of mergers and acquisitions later, the Baxter vaccine is still not being developed: Baxter spun off its vaccine business, which was later bought by the company Shire, and Shire "does not have a Lyme disease vaccine in the pipeline,” a spokesperson said.
While Baxter decided to divest itself of vaccines, it tried an unorthodox way of selling the rights to its Lyme vaccine, Plotkin said: Instead of negotiating a royalty agreement, which would be the industry standard for a product that has not yet been approved by the FDA, Baxter asked for a very large up-front payment. No one bit.
“Can you imagine my frustration that a vaccine I worked on as a young scientist isn’t out there helping people?”
Sam Telford, Tufts University
Years later, with little progress on this vaccine, Plotkin’s frustration is palpable.
“I was and am livid about the failure and stupidity and cupidity and anti-public health attitude of an organization to obstruct the use of their technology,” he said. “What makes it even sadder was that their data looked good when they decided to drop it,” he added.
Telford sees both a scientific and public health tragedy. “Can you imagine my frustration that a vaccine I worked on as a young scientist isn’t out there helping people?” he asked.
Telford recently wrote a grant to try to resurrect the technology behind LYMErix. In May, he found out it would not be funded.
It will be years before a new Lyme vaccine could make it through FDA trials. The next-closest Lyme vaccine, being produced by Valneva, a European biotech company, only announced its Phase 1 safety testing this past December. That means we are at least three to five years -- and hundreds of millions of dollars of investment -- removed from a Lyme vaccine, assuming that Valneva continues to develop it.
An Alternative Approach
Mark Klempner, a professor at UMass Medical School, is working on an alternative called “pre-exposure prophylaxis,” or PrEP.
PrEP sounds like a vaccine, but works differently. (See the graphic below.) It bypasses the vaccination process, which exposes us to some piece of the bacteria to build up our immunity against it, usually in the form of antibodies. Klempner has produced ready-made antibodies against Lyme that could be given in a once-a-year treatment. Then, when a tick bites, the antibodies would bind to the Lyme bacteria, preventing infection before it starts.
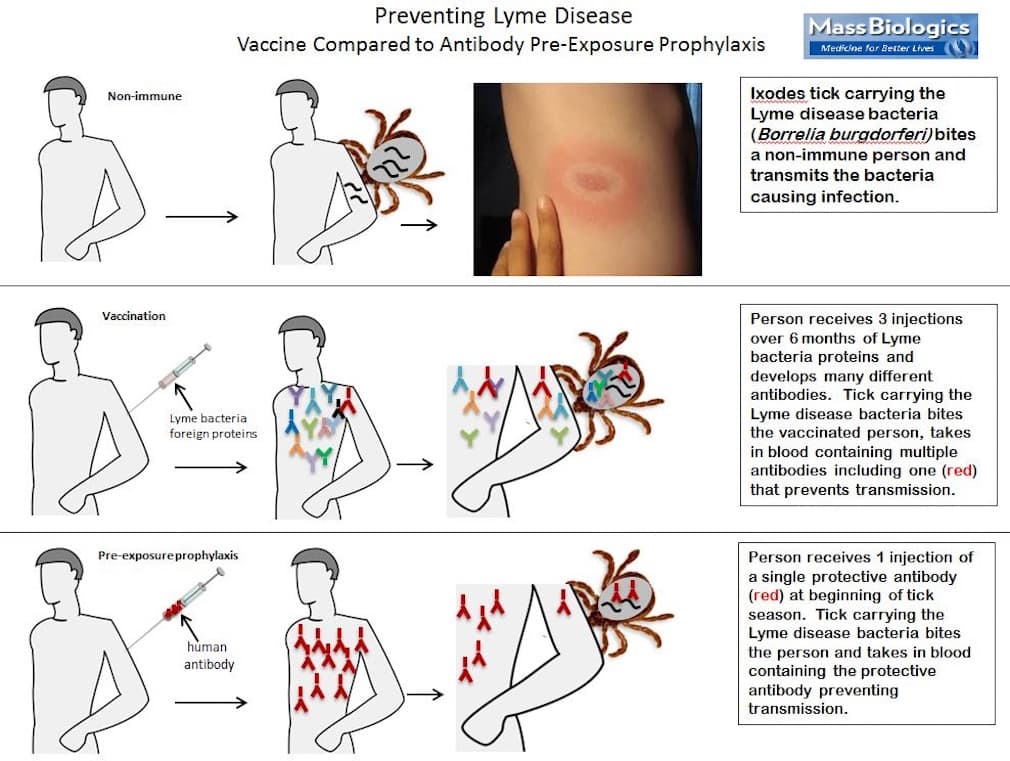
So far, PrEP has been 100 percent effective in preventing Lyme disease in animals. The next step is clinical trials, which again will cost millions of dollars. Klempner is worried that partnering with a pharmaceutical company will mean the PrEP price tag will put it out of reach of most people.
"We went to Big Pharma and we said, 'We're interested in moving this forward, we'll license it to you,' " he told a briefing at the Massachusetts State House this week. "And they said, 'Very interesting, we like the idea a lot. We're going to model it to cost $600 a dose, because we need to get our profit.' And we said no."
That's just not affordable, Klempner said, but if the antibody could be developed with state or federal backing — he's currently seeking $3 million to get it to the point of clinical trials — the price could be kept far lower, at around the LYMErix level of $210.
State Rep. James Cantwell of Marshfield and Scituate hosted Klempner's State House briefing, and said he was encouraged by the scientific progress on the antibody, and supportive of the effort to find public funds to develop it.
"This is one of these public health issues that we see as an absolute priority, because it is a public health crisis," Cantwell said. "It really is the largest public health crisis, I think, that we all see," and "something that every one of us is dealing with."
Klempner estimates that "if all the stars align perfectly," the antibody could be available to the public in three to five years. Because it is an antibody rather than a vaccine, it raises a lower level of concern about safety, he said.
Vaccine Dreaming
In the meantime, Plotkin hopes for an ideal vaccine that would work for both sides of the Atlantic, one that has few side effects, can be licensed for use in children, and protects at least 80 percent of those who get it for two years or more.
A vaccine like that might be “asking for the moon,” said Telford. A vaccine that worked less well would still be important: “Even reducing the risk 50 percent in New England would be fantastic,” he said.
But a vaccine won’t be a panacea. Most Lyme experts see a vaccine as just one part of a comprehensive strategy against ticks, because a Lyme vaccine won’t protect against other tick-borne diseases, which are on the rise.
“We need to fill our quiver of arrows as much as we can, give people a choice of things that we can be doing,” Telford said. “A vaccine is another arrow that people can take out of the quiver.”
Plotkin’s son, Alec, needed a pacemaker for a slow heartbeat caused by Lyme. But he got the necessary intravenous antibiotics and has fully recovered.
Still, his son's Lyme ordeal added impetus to Plotkin's push for a Lyme vaccine. “I had taken this point of view before his incident,” he said. “That reinforced it.”
With reporting by WBUR's Carey Goldberg. You can hear a conversation between her and WBUR's Morning Edition host Bob Oakes atop this post.
David Scales MD, Ph.D., is a physician at Cambridge Health Alliance and Harvard Medical School. He can be found on Twitter @davidascales.
This segment aired on July 24, 2017.
