Advertisement
Medical Milestone In Boston: 13-Year-Old Gets New Gene Therapy For Blindness
Resume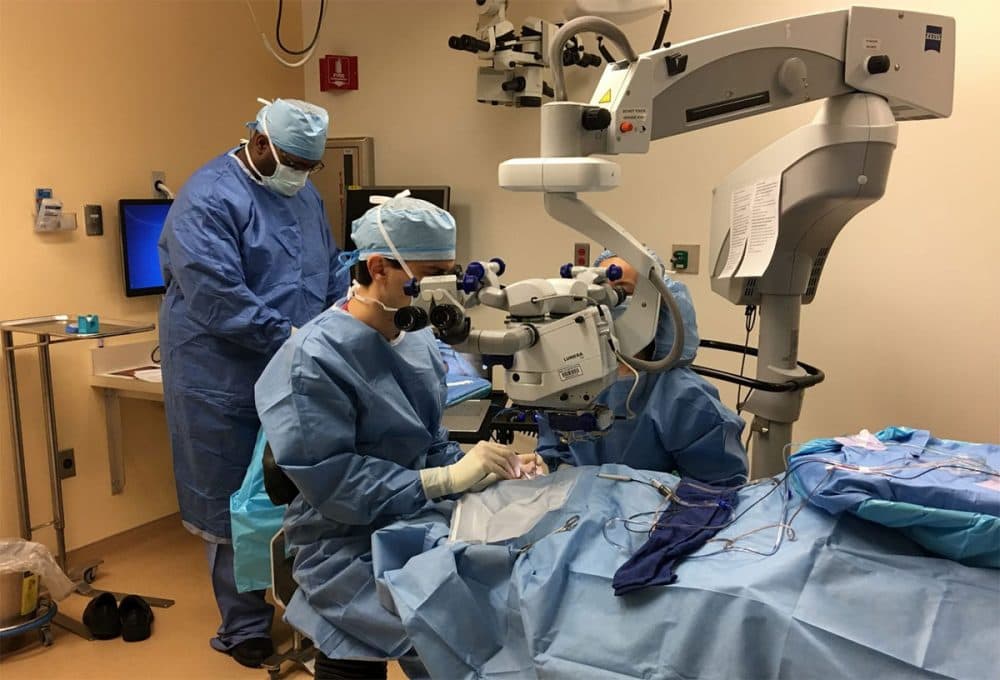
When Jack Hogan was 2, his parents noticed that he would walk into walls at night. Twice he even fell down stairs, says his mother, Jeanette Hogan.
She would tell him to pick up his toys, "And he wouldn't pick them up. And I'd be like, 'It's right there!' And he wouldn't see it."
Jack was ultimately diagnosed with a form of vision impairment that comes from lacking a gene that's key to how the retina, at the back of the eye, senses light. It gave him night blindness and left him lacking peripheral vision.
Now, at age 13, he has undergone surgery to try to improve his vision by fixing his genes. The procedure is an important medical first, and not just for blindness, says his Massachusetts Eye and Ear surgeon, Dr. Jason Comander.
"This is the first time that an FDA-approved gene therapy was given to a patient for any inherited disease," he says.
The gene therapy, called Luxturna, had been tried on more than two dozen patients in clinical trials, Comander says, and it improved most patients' vision either a little or a lot.
But Jack's surgery, after the FDA's approval last December, marks Luxturna's entry into the medical mainstream — including insurance coverage for its very hefty price tag: $425,000 for the eye that was operated on Tuesday, and just as much for the second eye, which is scheduled for next week.
Gene therapy works by packaging the needed genetic correction with a harmless virus that can deliver it into the patient's cells.
"The surgery had a very simple goal, which was to take three drops of liquid with this virus in it and put it underneath Jack's retina," Comander says.
The surgery itself was plenty complex, though — it involved removing some gel and membranes in the eye. Then, Comander says, "through a tiny little cannula, the tip of which is the size of a human hair, we inject these three drops of liquid underneath the retina. And inside that drop of liquid are many billions of copies of this virus. And the job of the virus is, it's very good at injecting DNA into cells."

Jack, normally active and athletic, had to lie on his back for six hours after the surgery Tuesday. But by Wednesday morning, he was up to introducing himself to reporters: "Hi, my name is Jack Hogan, and I'm 13 years old, and I live in Fair Haven, New Jersey. I like to play sports, basketball — well I can't play baseball..."
It's expected to take up to a month to see what effects the surgery has on his vision. Jack says he can't see a difference yet, but he has high hopes: "I hope it will increase my daylight — so I could see at night for longer," he says. "And peripheral vision, so I could play basketball, I could see a whole court."
An estimated 1,000 to 2,000 Americans have the type of genetic blindness that Luxturna treats. Dr. Comander says he has several patients who are good candidates.
"It's every eye doctor's dream to take someone who has bad vision and give them the chance of getting better," he says.
But the significance of Jack's operation is far broader, Comander says. The advent of successful, FDA-approved gene therapy for patients like Jack has boosted gene therapy in general — a field that crashed in 1999 after a young patient in a clinical trial died. It has been revived by the many researchers who devoted their work to getting it right, Comander says, and now it holds promise for treating many diseases.
"Different forms of inherited retinal disease are the ones that are near and dear to my heart and are also moving through clinical trials," he says. "On a broader note, people are working on hemophilia and other diseases as well."
The high costs of gene therapy treatments like Luxturna do remain a major concern. And for Jack Hogan, there's one other, much smaller downside: His mother says that better vision will mean that he should be able to do more household chores.
For a far more detailed description of Jack Hogan's treatment, STAT describes it at length here.
This segment aired on March 21, 2018.
