Support WBUR
Birth Control Pill Of The Future May Only Need To Be Taken Once A Month
The experimental birth control pill would open in the stomach like a flower, slowly releasing hormones over the course of a month.
It’s new technology with a unique mechanism that the founders of Lyndra Therapeutics hope will ease the burden of current oral contraceptives, which are supposed to be taken at the same time every day.
The company, which is based in Watertown, received a $13 million grant from the Bill and Melinda Gates Foundation last month to develop the pill. It is one of over 30 startups launched by MIT professor Robert Langer.
That pill isn’t a reality yet. It’s currently under development as a multipurpose capsule that slows the release of medicine inside a patient's stomach. The company successfully used the capsule to create a long-acting anti-malaria pill.
Now, the startup is reworking the technology to deliver a once-monthly oral birth control pill. “The basic design and materials are generally the same,” Langer explains.
The capsule works by protecting the medicine inside from the stomach’s digestive acids, Langer says. Inside every pill is a polymer — a chain of repeating molecules --that contains the drug’s medicine. Lyndra’s polymer unfolds into a star shape inside a patient’s stomach, preventing it from entering the lower digestive tract longer than a daily drug. The actual medication sits inside of the polymer and is slowly released through small pores at the center of the star.
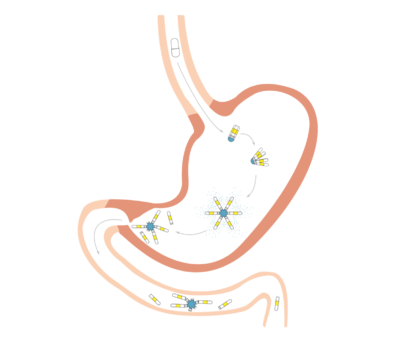
Some challenges remain with the monthly pill’s development, Langer says. While the capsule should in theory be able to deliver any drug, he says, the polymer must be customized for that particular drug.
“Anytime you have a new drug, there are different formulation issues,” Langer says. “Birth control is a very insoluble drug, so you might have to make [the polymer] more porous so that the release rates are right.”
Safety concerns arise when it comes to a monthly birth control pill like this, says Deborah Bartz, a professor of obstetrics, gynecology and reproductive biology at Harvard Medical School. Bartz is not associated with Lyndra.
Modern birth control pills have advanced to use a very small dose of estrogen to lower the risk of blood clotting. “Estrogen increases proteins in the body, including those that make blood clot,” Bartz says. But a monthly contraceptive might accidentally introduce too much of the hormone at once, she says.
Still, Lyndra’s pill could work, Bartz says, as long as the dosing of estrogen is controlled. “If this new pill slowly releases a lower amount of estrogen and progestin, there's no reason to think that the slower delivery of a birth control pill is any different than any other kind of pill,” she explains. “And having treated many, many patients, more options is always better.”
More options could mean fewer unplanned pregnancies. An estimated 44% of pregnancies across the globe are unintended -- a figure Lyndra CEO and co-founder Amy Schulman says is partly due to a lack of contraceptive access in developing countries.
“There are parts of the world where having an IUD or other forms of implantable birth control is either harder to access, harder to maintain or not really feasible given a lot of the situations that a lot of the women find themselves in,” Schulman says.
The Bill and Melinda Gates Foundation awarded the startup $13 million to improve contraception services to women in low- and middle-income countries. The technology could be a big help in certain environments where “it’s not always possible or desirable to take a daily pill,” Schulman says.
The Gates grant is specifically focused on creating a monthly contraceptive for the developing world, but Schulman has suggested that, if successful, her company would market the pill to women all over the world.
For physicians like Bartz in Lyndra’s own backyard, that goal is an exciting prospect.
"As long as the price point is good for this particular medication," Bartz says, "once it's developed it could potentially open up improved access for women within the U.S.”