Support WBUR
Coming Someday Soon: Your Own Implantable Medicine Cabinet
By Karen Weintraub
Guest Contributor
The thought of having to stick yourself with a needle is pretty unappealing, even if it’s to inject essential medicine.
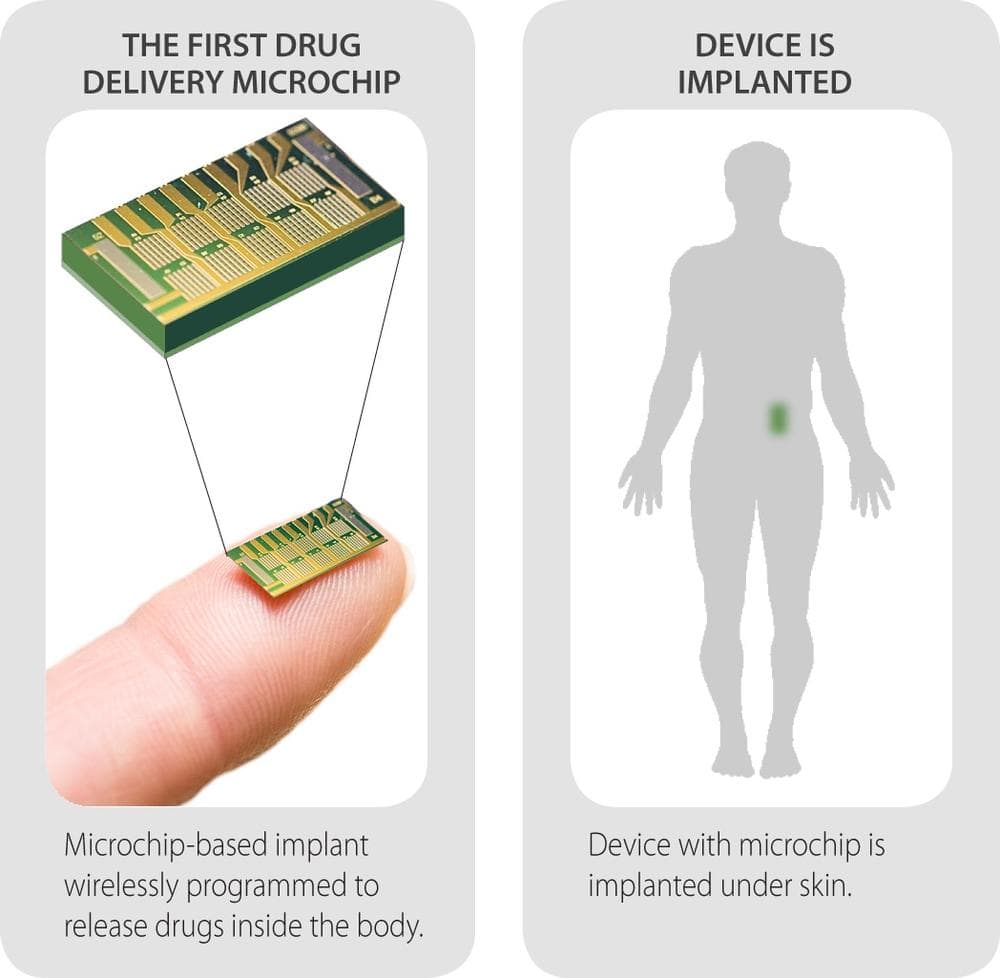
Now, researchers at MIT and a local biotech company have announced the development of an implantable device that delivers drugs from the inside. Think of Norplant - the now discredited birth control method implanted in the upper arm that was supposed to protect us from our own forgetfulness.
This device – which won’t be ready for patients for another four years at least – would be better, its developers say, because it would release a precise dose at a precise time.
About the size of a flash drive, the device would have 365 tiny compartments, potentially carrying different doses and different drugs. It could be wirelessly reprogrammed by a doctor if the dose needed to be changed.
“That really translates into freeing patients from the burden of managing their disease on a daily basis,” says Robert Farra, president, and chief operating officer MicroCHIPS, Inc., the Bedford company that is developing the device. Patients would “no longer have to remember [to take their medications] or deal with pain of injection.”
The company announced results today of a new study in seven patients taking the injectable osteoporosis drug Teriparatide (the drug-injection combination is sold in the U.S. under the trade name Forteo). All seven did fine as the device was implanted beneath the skin near their waist in a 20-minute office procedure using local anesthesia. They all participated through the full study and, after it was removed, all said they would consider getting the implant again.
Although all the patients developed scar tissue around the device, as was expected, enough of the drug was able to get through that tissue to help the patients as much as with FORTEO, according to the study, published in the journal Science Translational Medicine.
The device is based on the research of MIT invention dynamo Robert Langer (he holds roughly 800 patents or patents pending), and Michael Cima, an engineering professor at the Koch Institute for Integrative Cancer Research, among others.
If it passes remaining scientific hurdles and reaches the market, Farra says it would cost about the same as Forteo, roughly $10,000 a year.
Right now, Farra says there’s not much competition in the implantable drug delivery world.
“We’re not aware of other people with microchips used to deliver drugs,” he said.
But, not surprisingly, he thinks his product is a game-changer.
“This does trigger the age of telemedicine,” he said, potentially allowing devices to sense impending health problems and deliver drugs to cut them off. “It is a little bit on the science fiction side.”
In addition to osteoporosis drugs, the device could be used to deliver other drugs made of tiny proteins, like the interferon commonly used to treat multiple sclerosis, he said. Medications like the insulin needed by diabetics aren’t concentrated enough and wouldn’t fit inside the tiny device, he said.
Multiple sclerosis patients have a growing number of drug options, but some could certainly benefit from a device that would allow them to skip daily injections, said Dr. Nicholas LaRocca, vice president of healthcare delivery and policy research at the National MS Society.
“I would imagine that people would be willing to try it,” LaRocca said.
He hasn’t seen any major problems with people unwilling to take MS medications because of fear of needles. It’s always harder, though, to stick to a drug that’s meant to prevent a problem rather than reduce symptoms.
MS patients would probably be most excited, he said, by medications now being developed that could be swallowed instead of delivered by either injection or a new implantable device.
And Tommy Stoddard, an MS patient from Somerville, said he can see one advantage of a daily injection over an implantable device: A sense of control.
Stoddard recently switched to a new medication that requires a once-a month infusion instead, and he rather misses the sense of accomplishment and control he got from the five minutes he spent injecting himself every day – a procedure he didn’t much mind.
“It was a routine in which I was focused on my health, did a good job and felt good about it,” he said.
Karen Weintraub is a Cambridge-based Health and Science journalist and frequent contributor to CommonHealth.
This program aired on February 16, 2012. The audio for this program is not available.
