Support WBUR
If A Tweaked Flatworm Can Suddenly Regrow Its Head, Why Can't You?
Then as the years pass, we lose that ability. But early on, we've got a bit of the amazing growability that Narnia showed in its Chronicles. (Remember how, when the world was new in The Magician's Nephew, an iron bar dropped upon the ground sprouted into a lamp-post, and candies into fruit trees?)
I learned that fascinating finger-tip fact from University of San Francisco assistant biology professor James Sikes (except the associations with Narnia were all mine) in connection with an even more fascinating set of papers just out in the journal Nature.
It was a very surprising finding that one gene can rescue a regeneration process that was lost millions of years ago.
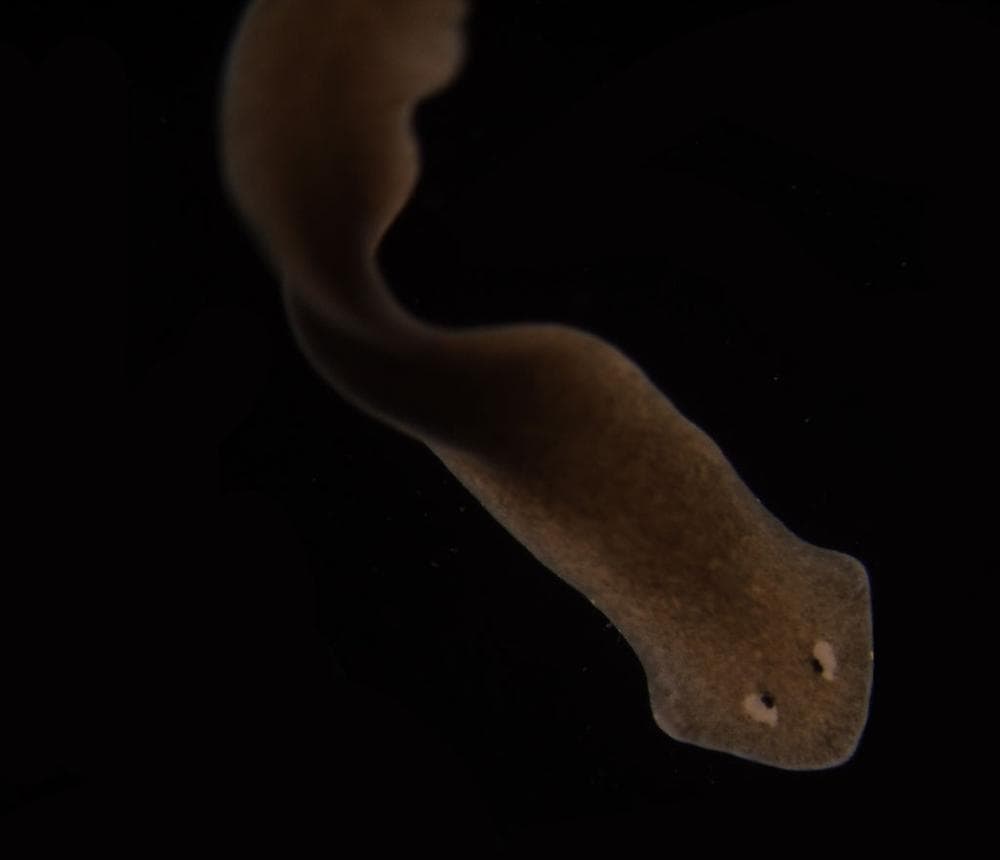
Allow me to sum up: Some of the worms known as flatworms or planarians are superstars at regeneration — "We can cut one worm into 250 pieces and have 250 worms a week later," Sikes said.
In particular, some flatworms can even regenerate their heads. Other planarians are not so good at the grow-back game. In the new research just out in Nature, scientists find that by tweaking just one gene's signaling pathway, they can restore the power of regeneration to those otherwise headless losers.
Is this surprising? You bet.
"I was shocked," Sikes said. "It was a very surprising finding that one gene can rescue a regeneration process that was lost millions of years ago."
I love this stuff. Here, lightly edited, is my chat with Prof. Sikes:
So tell me, please, why can I not regenerate my head?
[Tolerant laughter.] You want to try?
I might want to try if you'd let me know how to do it...!
It comes down to the issue that the regeneration ability is more limited, the more complex an animal is.
So complex animals don't regenerate very well. Whereas simpler animals like flatworms can regenerate amazingly well.
So there is some level of complexity there that we haven't figured out how to unlock. Our brains are much more complex than, say, a worm's brain, so at this point the best guess we have is that regeneration ability has been lost as we have evolved more and more complex structures.
So it's the price of complexity?
Exactly.
But then you have various species of planarians and some can regenerate and some can't. Why would that be?
Planarians are the winners in the animal kingdom for regeneration. We can cut one worm into 250 pieces and have 250 worms a week later. This is why we study them, because they're the best at it, so they're the ones to think about when you try to understand the process of regeneration.
But in some. we think there may have been a tradeoff between regeneration ability and some other fitness characteristic that these animals need to maintain.
Like reproduction?
We're hand-waving at this point when we have that in our paper, but it's a hypothesis. This [non-regenerating] species only has one shot at reproduction in its life, and it makes sense to maintain your reproductive structures as much as you can, and to keep them around so when it's time to reproduce you can do it.
But why that trade off, why would one preclude the other?
A little bit of background: Most planarians, the ones that regenerate really well, when they're amputated they tend to resorb all their reproductive structures so they have more energy to regenerate. So most planarians, if you chop off their head, their testes or ovaries will disappear and they'll give all their energy to regrowing that head.
And never get them back?
And then as soon as they’re done [with the head], they will regenerate [their reproductive organs], they will actually re-form them. So there is a time during the regeneration process when reproduction is precluded, these animals can’t do it at all. However the caveat there is these guys can reproduce throughout their life, they have multiple reproductive events throughout their lives.
Whereas this other species, the species that I looked at, is what we call semelparous, meaning it can only reproduce once in its life at the end of its life cycle. The key is that when these guys are amputated, we don’t see resorption of the testes or ovaries. So we’re wondering — again, this is handwaving, but our hypothesis is that maybe this aberrant signaling that we discovered that inhibits regeneration might be involved in maintaining [those organs] so that these guys can reproduce.
My big takeaway from the clump of papers that are coming out in Nature is that it is extremely striking that if you just tweak a 'signaling pathway' of a gene known as "Wnt," you can in fact enable regeneration. Was that a surprise and might it have some potential human medical application?
I was shocked. It was a very surprising finding that one gene can rescue a regeneration process that was lost millions of years ago. A million years ago is not that much in evolutionary time, but these guys have never regrown their heads in this tissue in millions of years, yet they maintained that latent ability to regenerate is in their tissues.
Which I think is the takeaway message from our paper: That even though an animal might not be able to regenerate in parts of its body, those parts of its body may still have the potential to do it if we can tweak the signals correctly.
I have to stop you there. Is there any chance that humans can have some latent ability to regenerate?
We think, sure. We have some potential to regenerate now. One thing people don’t realize is that children under the ages of 2 to 3 can regenerate their entire fingertips. It only last for a couple years after birth but you can regenerate new bone, new nerve, new blood vessels and everything, but then that ability disappears.
So it kind of gives us the hope and the hint that humans do have that latent ability. It does go away as we grow up, but that potential may still be lying in our body somewhere. And unlocking it is not going to be as easy as tweaking one gene, as it is in planarians.
But the take-home message is that for the first time, we can see that animals, three different species in three different papers, have shown that we can unlock it very, very easily in a simple animal. But can it be unlocked in a complex animal? That is the question.
Well, it would certainly be incredible if we could do some genetic engineering and tweak ourselves to be able to regrow our heads, but what about some less ambitious medical application?
At this point, the hope for regenerative medicine is mostly at the tissue level. We’re not going to be able to regenerate heads anytime, probably in the history of man.
But I think nerve regeneration, the fact that these guys can regrow complete brains by tweaking a single signaling pathway, is important. Can we unlock that potential for nerve regeneration, can we unlock the potential for something that's not as complex as an entire head, but is the potential there for us to do it on a smaller scale?
Why can our skin and our liver regenerate whereas our heart and our nerves cannot? That's the question: Parts of our body can do it but parts of our body cannot. So unlocking the potential in those specific parts does have the potential for regenerative medicine to play huge roles in the future.
Do we humans have a version of the "Wnt" gene you tweaked?
Yes, Wnts are involved in a plethora of developmental processes from sponges all the way to humans. We use Wnts all the time during embryogenesis, so when we’re developing as embryos the Wnts are on for a variety of different processes, and they’re just recycled over and over. Planarians use it to identify what is head and what is tail.
Has anyone tried tweaking them in humans?
Well, thats unethical. But we have looked at in vertebrates. We do see Wnt signaling alterations that are happening in vertebrate embryogenesis, and we do alter them and we alter embryonic development. We haven’t really looked a lot at post-embryonic development, things like regeneration.
Again, its not going to be a single signal pathway in vertebrates. It's going to be a little more complex than that. But yes, we have the Wnt signals, we have the same genes.
Readers, I don't dare ask you which organs you'd most want to be able to regrow. But if this work triggers a sci-fi flight of imagination in you, please share...
Here's the Nature press release:
Molecular cues that enable flatworms to regrow heads are identified in three independent studies published in Nature this week. By comparing planarian species that have full regeneration with species that have partial regeneration, the groups are able to uncover what it is exactly that allows regeneration to happen. They all show that tweaking a specific signalling pathway can promote regrowth of heads in planarians that have limited regenerative capacities.
Planarians are flatworms that are known for their regenerative capabilities, but whereas some species can regenerate a whole individual from a head or tail fragment, others can only regenerate tails. Yoshihiko Umesono and co-workers show that interplay between two signalling pathways (ERK and Wnt/beta-catenin) creates a gradient that selects for regrowth of either a head or a tail, depending on where the injury lies. They suggest that the balance between these signalling pathways may vary among planarian species, explaining why some species cannot regenerate heads. They demonstrate that Wnt/beta-catenin signalling interferes with head regeneration by showing that inhibition of this signalling activity enables head regrowth in a planarian species that otherwise cannot regenerate heads.
Similar conclusions are made in two separate investigations performed by James Sikes and Phillip Newmark, and Jochen Rink and colleagues. Sikes and Newmark find that Wnt signalling is aberrantly regulated in regeneration-deficient tissues. Both groups show that down-regulation of Wnt signalling in these regions restores regenerative abilities, including the formation of new heads. Together, all three studies reveal that manipulating a single signalling pathway can reverse the evolutionary loss of regenerative potential.
This program aired on July 24, 2013. The audio for this program is not available.
