Support WBUR
New Addiction Treatment Implant Will Hit The Market Next Month At $4,950
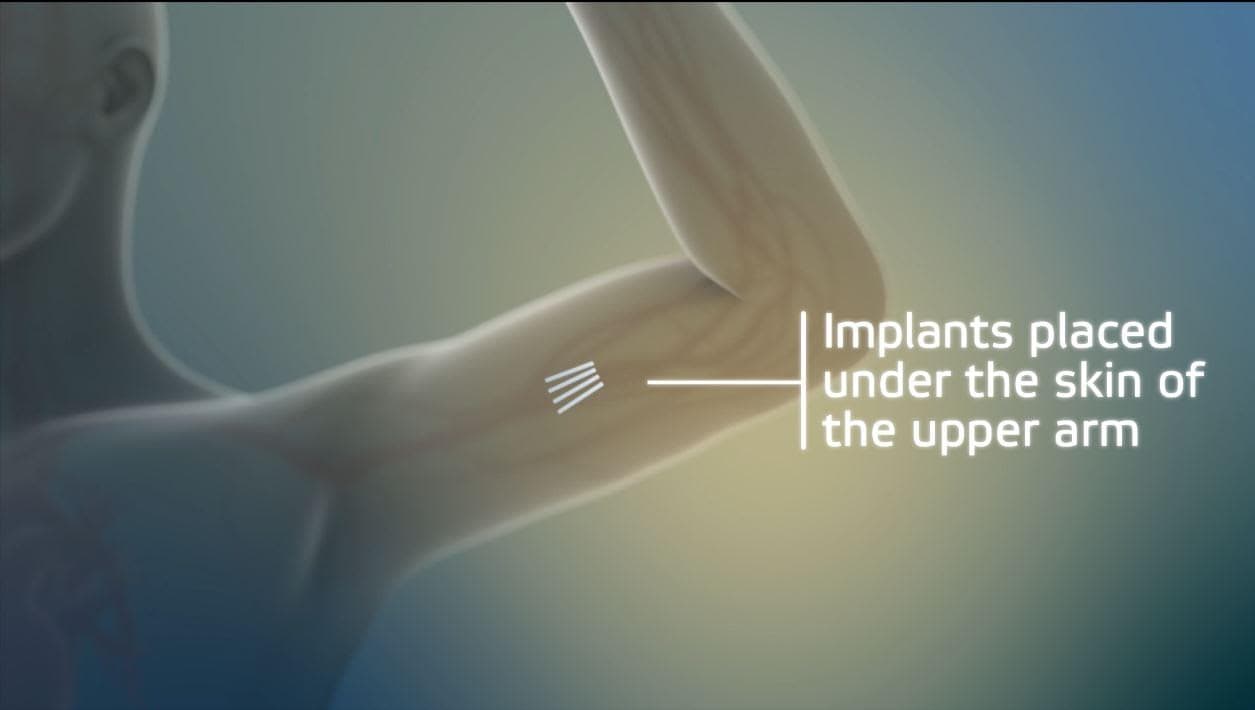
There's a new tool in the fight against the nation's raging opioid epidemic.
The FDA on Thursday approved an implantable version of the drug buprenorphine, which staves off opioid cravings. Labels for the new device are rolling off printing devices today, and trainings begin Saturday for doctors who want to learn to insert the four matchstick size rods into patients.
The implant, called Probuphine, is expected to be available by the end of June.
“This is just the starting point for us to continue to fight for the cause of patients with opioid addiction,” said Braeburn Pharmaceuticals CEO Behshad Sheldon.
But one day after the FDA approved this first long-acting delivery method for buprenorphine, debate continues about how effective the implant will be and whether insurers will cover it.
A Game Changer ... Or Set Up For Failure?
The head of the National Institute on Drug Abuse calls the new implant a game-changer because it will help addiction patients stay on their meds while their brain circuits recover from the ravages of drug use. And addiction experts say it will be much harder for patients prescribed the implant to sell their medication on the street, which is a problem for addiction patients prescribed pills.
"I think it’s fantastic news,” said Dr. Sarah Wakeman, medical director of the Substance Use Disorder Initiative at Massachusetts General Hospital. “We need as many tools in the toolbox as possible to deal with the opioid epidemic.”
Wakeman, however, is concerned that the implant only delivers one dose — 8 milligrams of buprenorphine. She prescribes between 4 and 24 milligrams of buprenorphine pills, depending on how much a patient needs to fight opioid cravings.
“This is a wonderful tool for someone who doesn’t want to take a daily medication” or someone who can’t manage doing so, Wakeman said. “If you need to add daily medication on top of Probuphine, you lose the added benefit that would come with not needing that daily pill.”
Sheldon says Braeburn Pharmaceuticals is testing weekly and monthly injections of buprenorphine that would be available in many doses.
Wakeman plans to sign up for a four hour Probuphine training, which includes a lecture, a demonstration, and practice inserting the implant. The company does not know yet if it will be safe to insert multiple implants into the same spot in the upper arm. A study on the efficacy is underway.
In the meantime, some doctors say they will hold off on using the implant. Dr. Indra Cidambi, who treats addiction patients in New Jersey, says she’s worried patients will assume it’s enough, that they don’t need check-ups or the counseling that is part of most recovery programs.
“Probuphine is set up for failure in that way,” Cidambi said, “because the patient will be seen after six months and in the meantime they’re not going to be following up with therapy and that means it’s not going to be medication assisted therapy. It is medication maintenance only.”
Braeburn Pharmaceuticals and the FDA say they expect patients to be in counseling while prescribed the implant.
Insurers Not Sure Implant Is Worth The Cost
Blue Cross, the state's largest health insurer, says it will cover the device, which will cost $4,950 and last six months — or about $825 a month. But some other insurers say they aren't sure yet if the buprenorphine implant is worth the price compared to the buprenorphine pills, which cost $130 to $190 a month.
“Certainly the drug holds great promise for individuals struggling with opioid addiction. However there’s still a lot we don’t know about its effectiveness,” said Eric Linzer, senior vice president for the Massachusetts Association of Health Plans.
Braeburn CEO Sheldon says that the implant will be cheaper than Vivitrol, another long-lasting treatment on the market. Vivitrol is a form of naltrexone that is injected once a month and costs about $1,000 a month.
Braeburn Pharmaceuticals says it may refund money to insurers if the Probuphine implant doesn't work to keep patients from relapsing, and offer rebates for patients who have to buy it on their own.
