Support WBUR
New Gene Therapy Shows Promise For Patients With Sickle Cell Disease

Lying propped up in a hospital bed and clutching a New England Patriots cap, Brunel Etienne Jr.’s face lights up when he remembers last month’s Super Bowl.
He screamed himself hoarse in Atlanta, Etienne says, as a nurse fusses over him and a machine slowly replaces his misshapen blood cells with someone else’s healthy ones. Patriots safety Devin McCourty had surprised him with free tickets.

“I was so happy. I was like in shock,” says Etienne, 26, who met McCourty several years ago at the hospital.
For Etienne, looking back is easier right now than looking ahead. The substitute cafeteria worker from Brockton will spend the next three weeks or so in an isolation room as chemotherapy destroys his blood cells.
If all works according to plan, that misery will be justified by the results: a cure for the sickle cell disease that triggered a stroke when he was a toddler and left him needing these monthly blood transfusions ever since.
“I'm tired of it,” he says.
Etienne volunteered for an innovative gene therapy trial at Dana-Farber/Boston Children’s Hospital aimed at combating sickle cell. If it succeeds, he won’t need any more treatments and won’t suffer any more sickle cell symptoms, like the exhaustion that sometimes makes it hard for him to live a normal life.
“When I was first introduced to the trial, I honestly jumped at the chance for it,” he says. “Even if it doesn't work, the research they have from me… [could help] kids who have the same disease.”
Sickle cell disease affects an estimated 100,000 Americans, many of them African-American, and millions more worldwide. It comes from a mutated gene that patients inherit from both their mother and father.
Instead of having all round, flexible red blood cells, sickle cell patients make pointy, rigid ones. These can get stuck in tiny capillaries and blood vessels, depriving parts of the body of oxygen and triggering symptoms – which can include pain, strokes, organ failure and even early death.
The disease can be cured with a bone marrow transplant , but the donor has to be a good match or the patient’s body will reject the new blood. Most patients, including Etienne, don’t have family members who are good match.
The new approach uses gene therapy to provide the same benefits as a bone marrow transplant, without the risk of rejection – because the patient gets back their own blood, says David Williams, chief scientific officer of Boston Children’s, who is leading the trial.
A Therapy More Than A Decade In The Making
It’s long been known that a fetus and newborn produce a different kind of red blood cell than an older child. Even newborns with the sickle cell gene make healthy blood cells, thanks to fetal hemoglobin, which allows a fetus to get enough oxygen from its mother’s blood. But the body usually stops making it a few months after birth.
About a dozen years ago, researchers at Dana-Farber/Boston Children’s identified the gene that controls the manufacturing of fetal hemoglobin, called BCL11A. If they could just keep this gene from shutting off production of fetal hemoglobin, they reasoned, they could prevent most or all of the symptoms of sickle cell.
“There was a lot of skepticism about whether this would work,” says Vijay Sankaran, a member of the pediatric hematology and oncology team at Dana-Farber/Boston Children's, who was involved in the gene discovery.
“For me, it's given an important lesson, which is: when you have an interesting finding, stick with it, try to push it forward and really focus on what the basic science can teach us,” Sankaran says.
The tricky part was figuring out how to make the genetic fix to every red blood cell, and not to other cells where it might cause side effects, Williams says.
Through years of experimentation, the researchers learned how to infect the right cells with a virus that inserts the genetic fix. Once the cells are edited, they can be infused back into the body of a patient whose own red blood cells have been killed with chemotherapy. Then, they should start making enough fetal hemoglobin to prevent problems from sickle cell.
The First Patient
Manny Johnson volunteered to become the first person ever to receive this approach to gene therapy for sickle cell.
He says he opted into the experimental trial because he has a 7-year-old brother who also has the disease. And though he’s generally healthy, like Etienne, he was sick of the transfusions he’s needed since he had a massive stroke at age 4.
“I kept telling myself, I have the least amount of problems with sickle cell, so I feel like I was more than well enough physically and mentally to do it. So, I was like: I should just go for it,” says Johnson, 21. “I did it for my little brother first, me second and then people – you know, the rest of the world.”
Johnson says he didn’t get bored during his month of isolation in the hospital. He just played a lot of video games.
But the chemotherapy was tough.
“Not sleeping, not drinking, losing your hair, skin – you know, everything,” he says. “But it was all temporary. That's why I was okay with it.”
That was last May. Since getting his gene-edited blood, Johnson has had no symptoms from sickle cell. He hasn’t needed any transfusions. And his blood, Williams says, looks nearly normal.
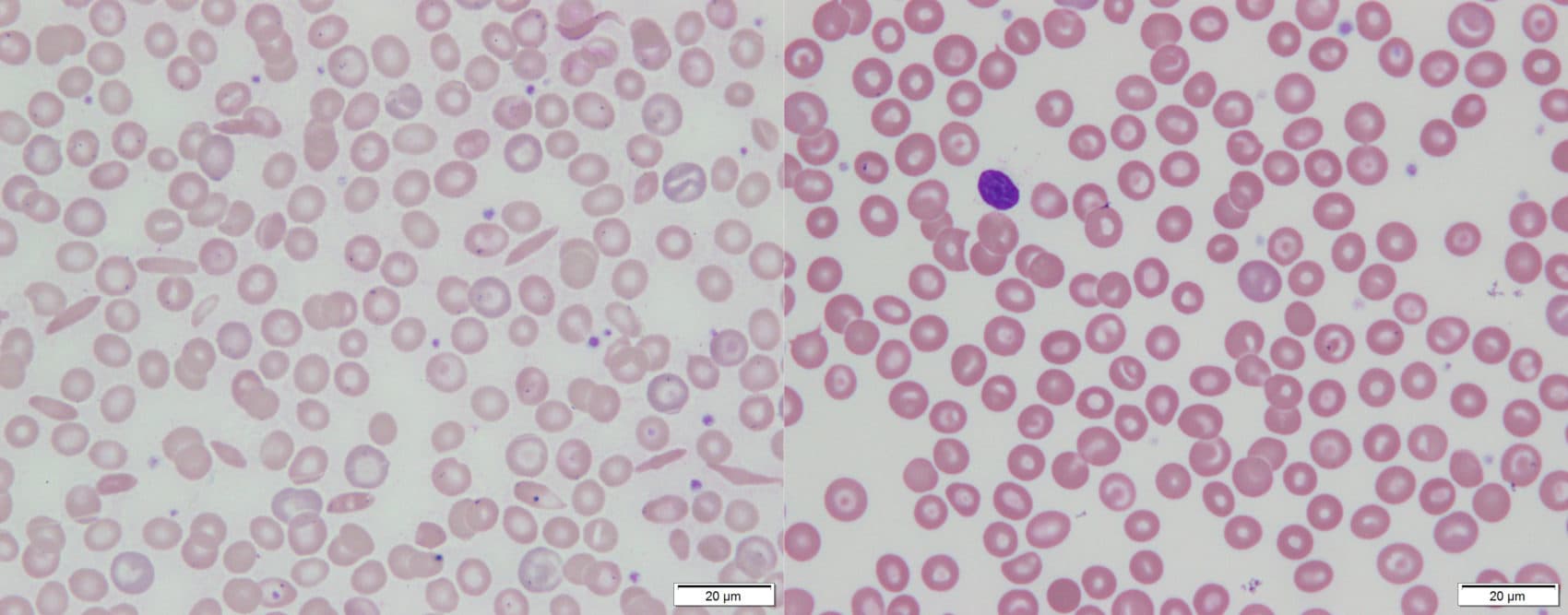
Johnson was in Williams’ office late last year when he saw pictures of his own blood for the first time.
“That's my stuff?” Johnson asked.
“That's your blood. From many years ago,” Williams responded, pointing out the sickle-shaped cells that dot the slide. Then he gestured toward a second image from after Johnson’s gene treatment. “There’s no sickle cells we can find,” Williams said.
Sickle cell disease is very expensive to treat. One study estimated it at nearly $1 million across a lifetime, plus another $700,000 in lost income for each patient. Although the one-time gene therapy doesn’t have a price tag yet, if approved by the U.S. Food and Drug Administration it’s expected to be extremely costly – perhaps akin to cancer therapies, intended as cures, that top a half million dollars. But Williams says gene therapy for sickle cell could justify a high cost, if it means the end of future medical expenses, not to mention the pain and suffering of the disease. He hopes that eventually an inexpensive pill will be able to yield the same relief.
Williams will continue to follow Johnson’s health, but he says the results so far are encouraging. He’s looking to repeat Johnson’s success with other patients. “We want to prove that it is not just him, that it's in every patient and we want to prove it's durable,” Williams says.
That’s why Etienne, patient number 3 in the trial, is heading into the hospital this weekend.
But instead of thinking about the treatment he’s about to receive, Etienne is still thinking about the Super Bowl – and his new goal of going back next year. Hopefully by then, he won’t have to worry about running out of energy, or rushing back to Boston to get another blood transfusion.
“We're putting money away little by little,” Etienne says of himself and his mom, who went with him to Atlanta. “Next one's going to be in Florida.”
This post has been updated.
This article was originally published on March 08, 2019.
This segment aired on March 8, 2019.
