Support WBUR
Commentary
Hope is its own form of healing

Imagine having a toddler who starts missing his developmental milestones. Then imagine sitting across the desk from a doctor who is about to share the results from a battery of tests ordered because of your worries. You still think there’s a decent chance the doctor could say there’s nothing wrong with your beautiful little boy. You still hope he’s simply a late bloomer. Instead, you’re handed his death sentence: Duchenne muscular dystrophy (DMD), a ruthless neuromuscular disease that impacts one out of every 3,500 boys worldwide. The condition begins robbing victims of muscle in utero and culminates with cardiac and respiratory failure in young adulthood. Patients are typically wheelchair-bound by early adolescence.
Hearing those words is nightmare enough, but the rest of the standard diagnosis speech doubles down on heartache. “There’s nothing we can do,” parents have been told. “Just go home and love him.”
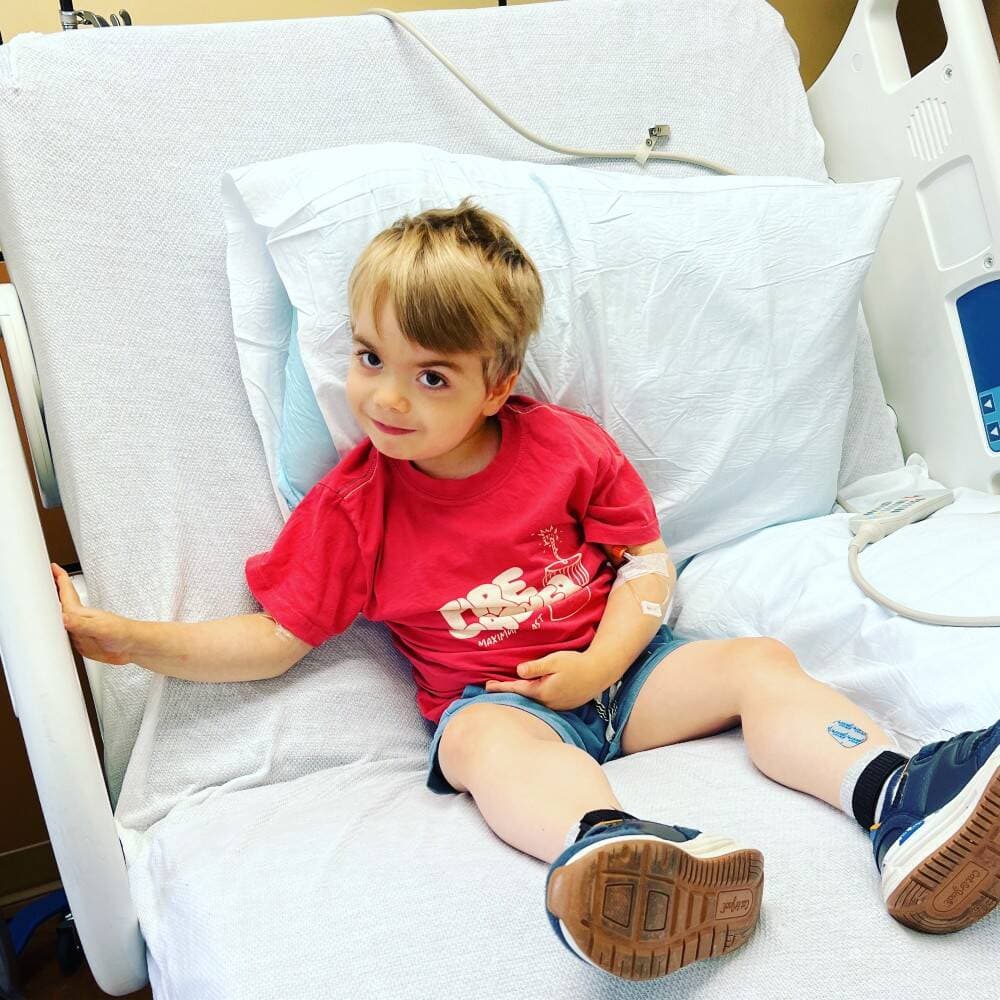
I know, because I am one of those parents. But when my son received that diagnosis on a cloudy fall afternoon in 2020, the outlook had brightened ever so slightly from decades past. I first heard of muscular dystrophy when my parents turned on the annual Jerry Lewis MDA Labor Day Telethon in the 1980s. Little did I know then how important the science happening behind that telecast would become to my family all these years later. I couldn’t have understood that the promise of gene therapy, unimaginable back then, would mean the difference between “Go home and love him,” and “There is hope.”
Hope, it turns out, is its own form of healing. When my son qualified for a clinical trial of Sarepta’s SRP-9001 gene therapy and received his first infusion in the phase 3 double-blind study last year, I took my first deep breath since he was diagnosed. Mercifully, each patient will receive the gene therapy by the end of the trial. And although it’s a two-year study and we aren’t sure whether he’s already received the active treatment yet, just knowing that we are doing something that might help him gives us palpable relief.
Until you’ve made the decision to enroll your child in an experimental trial of a next-generation treatment, you have little idea of the risk/reward calculus it requires. As many rare disease parents will tell you, it’s yeoman’s work — talking to experts, reading medical literature, attending webinars. Thinking, thinking, thinking. Praying, praying, praying. Ultimately, we decided that the certainty of a tragic outcome for our son, if we didn’t enroll him, would be worse than any potential risk associated with a relatively new treatment. We felt the evidence, both published and anecdotal, tilted the scales strongly in the therapy’s favor. We put all of our chips on the table of scientific progress, both for our own family and for others who might benefit later. And we haven’t second-guessed that decision for a single second.
Until you’ve made the decision to enroll your child in an experimental trial of a next-generation treatment, you have little idea of the risk/reward calculus it requires.
Now we are at an inflection point, where many other parents may — and should — have the opportunity to make the same choice for their children. On May 12, the U.S. Food and Drug Administration (FDA) will convene a committee of independent advisors to consider whether to recommend accelerated approval of SRP-9001, the therapy being tested in my son’s trial. This is the first time a gene therapy will be considered for accelerated approval based on evidence accumulated to date, even as the phase 3 trial continues to completion. Accelerated approval has enormous potential to write a new and promising chapter in the history of this relentless disease. If we enable a therapy to reach these boys’ muscles with a functional level of dystrophin, the protein their bodies lack, it could transform everyday life for them, their caregivers and their doctors — starting now. That’s why their stories are essential to the conversation on May 12 and beyond.
There has been and will continue to be intense scrutiny and debate over the treatment’s merits and each phase of trial results. Some decision-makers may take a wait-and-see stance, suggesting we delay approval until the phase 3 data is released. Others — namely the patients and caregivers who live with Duchenne’s cruel realities — will explain exactly why we don’t have that luxury.

The advisory committee has made some changes for this hearing that many in the Duchenne community say are antithetical to the committee's very purpose: to give patients and providers a voice. The committee has reduced the time allotted for public comment from 90 minutes to 60, and is using an unusual lottery approach to admit invited speakers, so some of the most informed witnesses will not be heard during the live hearing. And since cameras won’t be permitted for the virtual meeting — another change to the rules — patients and caregivers won’t be able to make eye contact, limiting their ability to form a human connection as they share the full scope of this treatment’s benefit, both potential and realized.
That is why it's imperative that the committee watch and absorb the videos from patients and health care providers that have been submitted as testimony on the public docket, and take them into full account when making a recommendation. The people who made these videos speak for patients on the cusp of losing their mobility and independence — the boys and young men who desperately want to stop this disease’s progression so they can continue to walk, eat, breathe and dream. They speak for parents, weary from the full-time job of caretaking and advocating, who hope that the government will grant the help that science offers. And they speak for health care providers and researchers, who are anxious for the fruits of their tireless work to be made available to every patient with Duchenne.
Listening to all of these voices is what committee members owe families like mine. Families who had the courage to participate in a trial and remain committed to participating in the scientific due diligence required beyond an accelerated approval.
We’ve done, and will continue to do, our part; now it’s up to the advisory committee and the FDA to help us realize a hopeful future for boys with Duchenne. Time is muscle, and we simply cannot afford to wait for tomorrow when help is here today.
