Advertisement
From The War On Cancer To The Moonshot: 'What's Different Now Is Everything'
Resume
This story is part of our "This Moment In Cancer" series.
Nearly a half-century separates the Obama administration's Cancer Moonshot from President Richard Nixon’s War on Cancer. But Greg Simon, the moonshot's inaugural director, says the two programs are really one marathon effort.
“We’re not starting a new war on cancer,” says Simon, a former corporate executive who has a type of blood cancer. “We’re trying to finish the one we’ve been in. What’s different now is everything.”
Simon reflects the general bullishness among cancer cognoscenti about the prospects for a great leap forward in cancer care. That fresh enthusiasm is why we're launching a series to capture the excitement that permeates cancer research labs and clinics in Boston — long an epicenter of cancer research.

"When Nixon's War on Cancer was launched," Simon says, “we had no army, we had no weapons, we had no strategy.”
But now, though no one expects a "cure for cancer" around the corner, scientists understand many of the molecular defects that drive healthy, normal cells to turn cancerous. That allows them to design drugs that target the defects and tame the cancer — at least in some patients, and for varying lengths of time.
Researchers also have powerful new tools to gather and sift mountains of scientific data. They can share that knowledge instantly and globally, accelerating the path from data to discovery.
The Biggest Buzz
But the biggest buzz is about immunotherapy. The idea that the immune system can recognize and attack cancer cells with exquisite precision is a century old. But only now are researchers finally learning to make it work — sometimes with spectacular results.
"There's more attention being paid to this area than any other today — in the research environment, in the clinical development environment, in pharmaceutical companies," says Tyler Jacks, director of MIT's Koch Institute for Integrative Cancer Research. "I’ve really never seen anything like it."
Probably the best-known success story is former President Jimmy Carter. He had melanoma that spread to his brain. But now, after treatment with immunotherapy along with radiation, he has no detectable cancer.
"There's more attention being paid to this area than any other today ... I’ve really never seen anything like it."
Tyler Jacks, director of MIT's Koch Institute for Integrative Cancer Research
Researchers are betting that immunotherapy, in combination with other treatments, will eventually vanquish many malignancies, including major killers such as lung and colon cancer. But many patients are already benefiting from the growing arsenal of treatments.
For example, meet Sheila Mulcahy. She was a 50-year-old oncology nurse on Cape Cod when she was diagnosed with chronic lymphocytic leukemia 11 years ago — the same type of cancer that afflicts Greg Simon. "My journey since then has been a complicated one," she says — an understatement.
One type of cancer caused tumors all over her body, including her eye sockets, requiring treatment that has left her blind in one eye. A second type of cancer forced surgeons to remove her stomach and gall bladder.

Doctors thought she'd die years ago. But not only is she alive, Mulcahy says she enjoys a "rich and colorful" life. Drawing on her nursing knowledge as well as personal experience, she now helps other cancer patients decide which treatments to get.
"The choices that were available to me 11 years ago compared to what they are now is phenomenal," Mulcahy says. "The research is great!"
Finding The Target
Mulcahy owes her life to “targeted therapy,” the fruit of research that has shown just what causes a well-behaved cell to go “renegade” — become a cancer cell. That process starts from damage to the DNA inside a cell that makes it race out of control, as though its accelerator pedal is stuck to the floor and its brakes are blown.
"Over the last two and a half decades, one has made drugs, which are highly targeted and specific, to try to cure some of these molecular defects inside cancer cells," says professor Bob Weinberg of MIT and The Whitehead Institute, who did some of that path-finding research. "Some of these drugs have proven to be brilliantly successful."
The drug that Mulcahy took for her form of leukemia is one of those. It has controlled her blood cancer with far fewer side effects than old-fashioned chemotherapy.
But precisely targeted drug therapy hasn’t worked for most cancers. Relatively few patients have benefited, and the effects often don’t last.
Dr. Vicki Jackson, chief of palliative care and geriatric medicine at Massachusetts General Hospital, says these unpredictable and patchy results are posing unprecedented challenges for many cancer patients.
"There is hope in a really new way, which is an incredible gift," Jackson says. "My patients know some of these medications [can] essentially melt the cancer away ... But they worry, if this medicine stops working, will there be another one? It's hard to live in that space."
"There is hope in a really new way, which is an incredible gift."
Dr. Vicki Jackson, chief of palliative care and geriatric medicine at MGH
Mulcahy, for one, is confident she'll have new escape routes if and when her current therapy stops working. "I think: Don't despair," she says. "There'll definitely be more options."
Brick Walls?
Immunotherapy might well come to her rescue. Last year, the Food and Drug Administration approved two such drugs for treatment of CLL, Mulcahy's diagnosis. But Weinberg, of MIT, says we don't know yet how far immunotherapy can go.
"A question is: Is the diversity of cancers that can be treated successfully by immunotherapy essentially unlimited as we begin to tweak these therapies? Or will we begin to hit brick walls?" Weinberg asks.
Those obstacles are important to keep in mind. The history of cancer treatment is full of shining promises and crushing disappointments. Radical mastectomies and bone marrow transplants were once touted as promising for breast cancer — mistakenly. Back in 1998, drugs to block tumors' blood supply prompted predictions that they would soon cure cancer. Wrong again.
The trouble is there will never be one “cure” for cancer. After all, it’s as many as 200 different diseases. And now each of these types is being divided into genetic subtypes that require specific treatments.
Moreover, even when cancers respond, most become resistant to treatments, because they mutate over time.
Dr. Nikhil Wagle, of Dana-Farber Cancer Institute, compares cancer treatment to the carnival game of whack-a-mole, where you smack a stuffed mole in one spot and it pops up in another.
"But if you know all of the different ways you need to whack a mole over time," Wagle says, "and develop a safe way to give all of those treatments in combination, like we do with HIV, then you basically whack all of the places that the mole might come up simultaneously, and then the mole can't come up anymore."
Wagle is talking about a growing consensus that the way forward in cancer has to come from combinations of treatments — adding immunotherapy and other new drugs to the chemo and surgery and radiation we already have.
Good News, Bad News And Budget-Busters
That’s the future — we can hope. Meanwhile, there has been big progress. This month, the American Cancer Society announced a 25 percent drop in cancer deaths over the past quarter century.
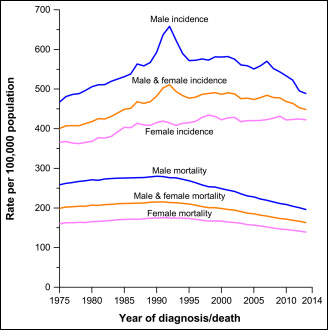
But the bad news is this: A quarter of Americans who now die of cancer shouldn’t. Dr. Otis Brawley, the Cancer Society’s medical director, says they asked what would happen if all cancer patients had a college education. The answer: "Instead of 600,000 people dying this year, if everybody had the death rate of college educated Americans, it would be 450,000."
That's because the best-educated Americans are much more likely to have healthier lifestyles and get preventive care as well as state-of-the-art treatment.
But if access to expensive cancer care is already a problem, new cancer treatments are real budget-busters — without exception.
Just take the latest and greatest new treatment for the kind of cancer that afflicts Sheila Mulcahy, chronic lymphocytic leukemia. It costs more than $100,000 a year — and patients need to be on it for the rest of their lives.
Dr. Jagpreet Chhatwal, at Massachusetts General Hospital, calculates that the new, more effective therapy will cause the overall cost of treating patients with that leukemia to balloon by nearly six times. That's due to both the cost of the new drug and the growing number of patients who will live longer and thus will need continuous treatment.
And this is only one relatively minor form of cancer. "We think this is just the tip of the iceberg," he says. "The trend is very disturbing. Clearly the sign is that treatment will become unaffordable in the near future." Chhatwal and his colleagues published their analysis earlier this month in the Journal of Clinical Oncology.
The urgency to solve this cost problem will only grow as researchers devise ever more effective ways of attacking more types of cancer.
Scientists are confident they can -- more confident than they’ve ever been. Although there’s no “cure” around the corner, the prospects for tomorrow’s cancer patients are definitely looking brighter.
Do you have a question about cancer research, treatment or care that'd you like us to investigate? Ask it here.
This segment aired on January 30, 2017.