Support WBUR
Boston's Weekly Health Newsletter
How a Mass. doctor helped engineer a promising new treatment for bladder cancer
Editor's Note: This is an excerpt from WBUR's weekly health newsletter, CommonHealth. If you like what you read and want it in your inbox, sign up here.
Bladder cancer hits men especially hard. The American Cancer Society estimates that doctors will diagnose about 85,000 new cases in the U.S. this year, about three-quarters of them in men. It’s the fourth most common cancer men experience, according to the National Cancer Institute.
And for half a century, treatment hasn’t really changed — until now.
For decades, if their cancer didn’t respond to chemotherapy or other existing treatments, most patients with the most common type of bladder cancer faced the same fate: a surgery to remove part or all of the bladder. Known as a cystectomy, it’s a life-altering operation that leaves many people with a small opening in the stomach, and an external urinary pouch for the rest of their lives.
“As a treating physician and a surgeon, I used to remove bladders for a living,” said Dr. Chris Cutie, a former urologist at Mass General Brigham. “It's a very onerous operation.”
Scientists have been trying to create new treatments for bladder cancer, but they kept running into a major hurdle. Keeping medicines in the bladder is well… kinda hard.
“The bladder is really good at evacuating things. It doesn't like to keep things in,” Cutie said. “Drugs stay in the body for about 30 minutes, which is just not a lot of time for drugs to work on these very complicated tumors.”
Cutie said drugs are typically injected directly into the patient's bladder. Then, to maximize exposure, doctors tell them to hang on for as long as they can before urinating and expelling the treatment.
“We’d have them watch ‘Judge Judy’ in the waiting room and say, ‘Hey, can you just make it to the next commercial before you empty your bladder?’” Cutie recalled.
Some doctors would even ask patients to roll around on the floor to coat the bladder with medication. The process was uncomfortable and, Cutie said, largely ineffective.
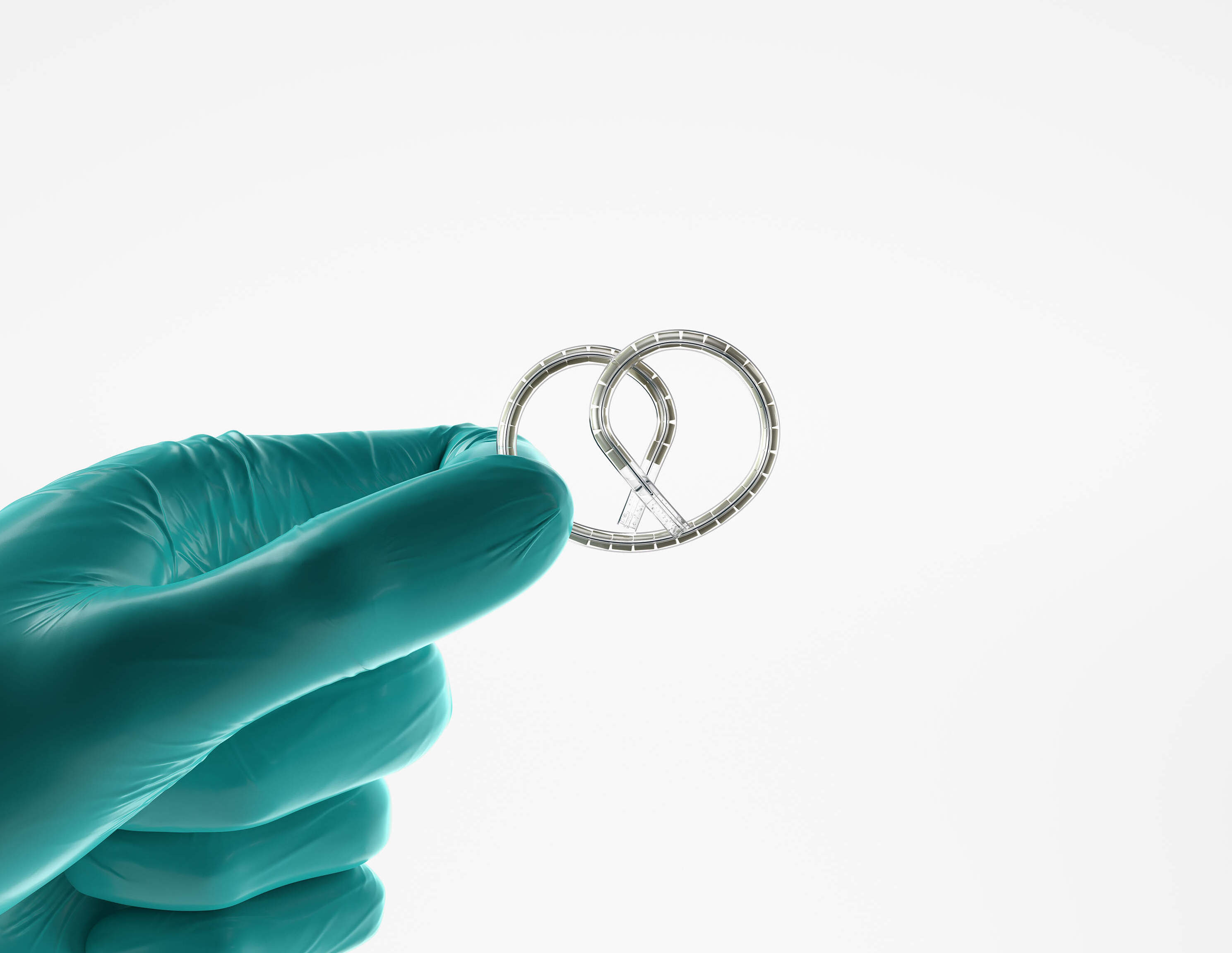
A promising pretzel
Cue a new treatment, approved by the FDA in September, that’s offering patients some hope. Called Inlexzo, it uses a pretzel-shaped implant to slowly dispense cancer-fighting medications.
In a small clinical trial, 82% of patients that got Inlexzo paired with a drug saw their cancer disappear, and more than half remained cancer free after at least a year, according to Johnson & Johnson, the company developing the treatment.
From his Boston-area lab, Cutie helped engineer the system with scientists from TARIS Biomedical, an offshoot of MIT, and Johnson & Johnson, where he’s now a vice president. He said this is the first real advance for patients with the most common form of bladder cancer in over 50 years.
“What we’re really proud of is that we see complete responses — meaning the disease is completely eradicated — within the first three months of treatment,” Cutie said.
For many patients in the trial, a cystectomy would have been the next step. Bill Parisi was thinking of retiring when he got his diagnosis in 2021. Parisi said he tried the typical treatments, but his doctors kept finding new cancer spots.
Soon, they told him it was time to remove his bladder. Parisi panicked.
“ I said, ‘Whoa, whoa, whoa.’ And ‘I don't like that,’” Parisi said. “I’m still running my business. If you take my bladder out, that's it. It would curtail my business.”
Parisi’s other option was surgery to rebuild his bladder, but he said it would have been difficult physically and disruptive to his life. His doctor suggested he participate in the clinical trial.

Parisi began treatments. He got the implant, then returned to have it refilled with medication every three weeks.
The treatment caused some irritation, but Parisi said keeping his bladder made the discomfort worth it. After 18 months of treatment, he was cancer-free and the implant was removed.
“ To me this is a lifesaver, it really was,” he said.
And seeing the evolution of the treatment makes him feel like “a hero” for helping to move the medical field forward — and hopefully saving other patients’ the pain of losing their bladders.
Long-term outlook
Dr. Adam Kibel, chair of urology at Mass General Brigham, said the trial results look promising, but it will take time to determine whether treatments like Inlexzo improve quality of life and survival rates for patients.
“This does a better job” of lengthening exposure to cancer-fighting drugs, said Kibel, who was not involved in the trial or developing the treatment. “The downside is someone has to put it in and out of your bladder.”
Kibel questions whether patients can get used to the idea of having something bouncing around in their bladders. He also cautioned that while this addresses one challenge to treatment, there are others, like finding the right drug for each patient.
“This is a complicated space with many new treatments coming up,” Kibel said. “We’ll see which ones appear to work best in terms of controlling cancer while minimizing side effects.”
Correction: An earlier version of this story incorrectly referred to the implant as a device. It was approved by the FDA as a drug. In addition, the company involved in its development is TARIS Biomedical, not TAURUS. We regret the errors.
