Support WBUR
Netflix’s ‘Joy’ tells the story of families like mine
In a scene from the new movie, “Joy,” streaming on Netflix, Bill Nighy — who plays the OB-GYN Patrick Steptoe, one-third of the British team who pioneered in vitro fertilization (IVF) in the 1960s and 1970s — tells a couple sitting across his desk that the then-experimental treatment probably won’t work for them.
“So why would we do it?” asks a prospective father.
“Because it might mean that others do have babies,” answers a would-be mother.
In our public discourse around scientific fact, even the merits of science itself, we can forget that there is an inherent, exquisite poetry to scientific inquiry and discovery — beautifully moving stories of personal persistence and courage contained within each and every step forward. That long before a blockbuster drug or novel intervention exists there is a researcher, a lab, patients and a problem to solve. That for all its rules and logic, medical science is, at its root, driven by our essential humanity — our innate, universal desire to thrive and progress, together.
“Joy” tells a story about one of the most evolutionary, biological instincts: to have a baby, to create a family. It is so primal a desire — and so fundamentally devastating when that desire can’t be fulfilled — that it’s somewhat remarkable to think it took until July 25, 1978 for science to finally offer a working solution for many common causes of infertility. Notwithstanding the unfortunate recent politicization of IVF, the treatment is so commonplace today (about 500,000 babies are born by IVF worldwide each year) that it takes a movie like “Joy” to remind us of a time when prospective parents with an infertility diagnosis had no options whatsoever.
Indeed, it took the singular team of Steptoe, the physiologist Robert Edwards and the previously unsung nurse and embryologist Jean Purdy to finally meet this essential human problem with translational science, resulting in the world’s first “test tube baby,” Louise Brown. A game-changing idea like IVF is just that — an idea — until an entire operation is built around it in order to bring it into the clinic to test its safety and efficacy. This requires careful study design, the development of protocols, enormous amounts of funding and regulatory sign-off. Most critically, it takes human volunteers — people with the courage to raise their hand and line up for what is, at that point in time, an experiment. In the case of IVF, it was entirely unclear whether it would work until willing women and men came forward to endure endless injections, surgeries and other indignities, fully aware that the realization of their own baby might remain out of reach.

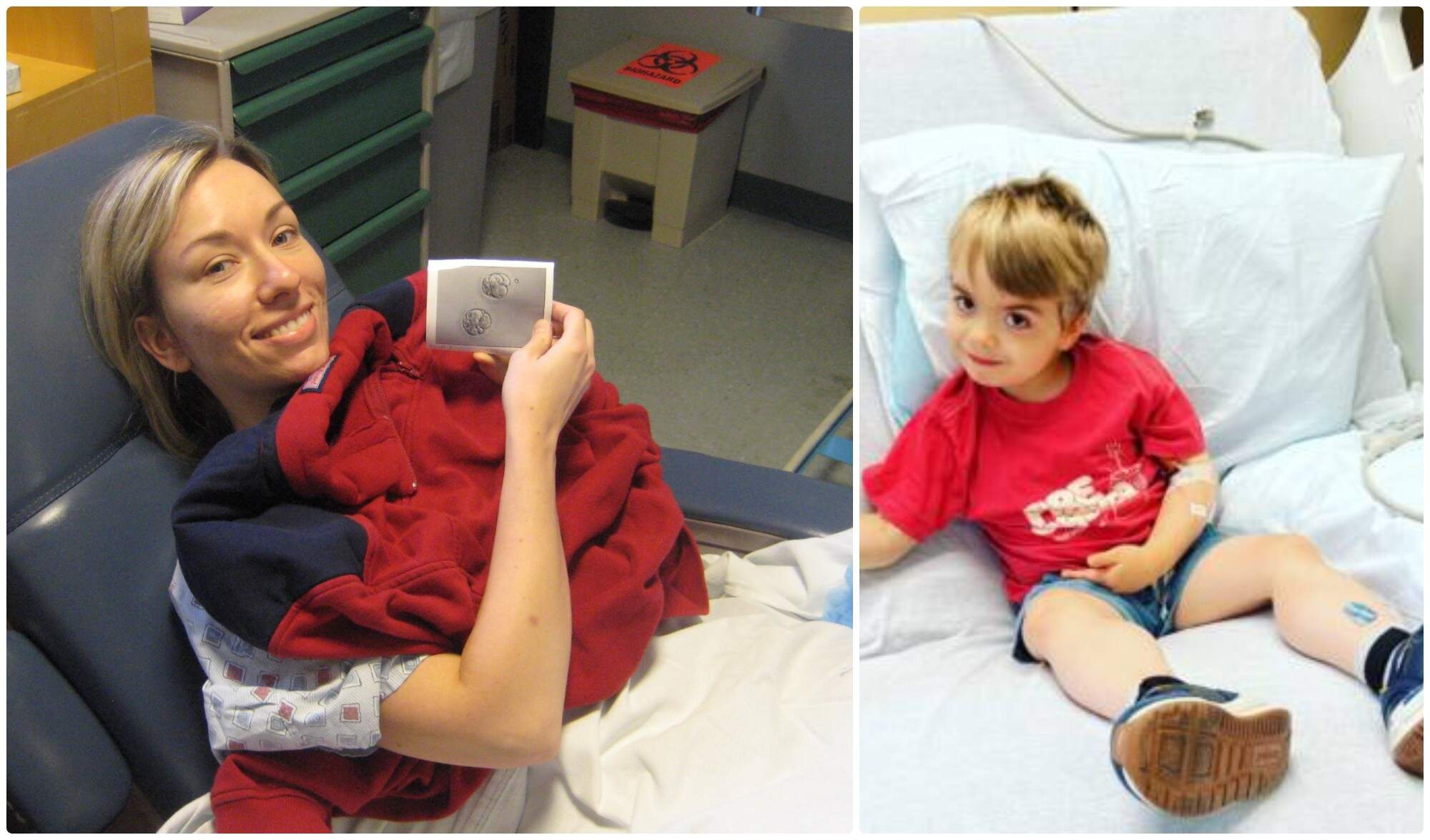
I am a beneficiary of that work and sacrifice. In 2010, with the help of a brilliant and compassionate doctor at Brigham and Women’s Hospital, I gave birth to my own IVF baby. Science made me a mother, and while nothing about the process felt particularly easy, natural or guaranteed, I never had to wonder whether it could work because Steptoe, Edwards, Purdy, baby Louise’s mother — Lesley Brown — and millions of other mothers before me had proven it did.
My second IVF baby, Charlie, enabled us to do our own part for medical science when he was diagnosed at age 3 with the rare and devastating condition Duchenne muscular dystrophy (DMD). As we had with infertility more than a decade earlier, in the face of this life-limiting diagnosis my husband and I turned immediately to science for solutions, for hope, to guide our steps forward.
A year after Charlie’s diagnosis, we learned that a potentially pivotal clinical trial was enrolling patients and that Charlie might be eligible to enroll. We weighed the decision with both heart and mind. It was not fully clear whether the experimental treatment would work for our son; early results were promising, but not definitive. Further, the therapy would be delivered to our son’s muscles via a high dose of a viral vector, thereby limiting his potential to receive other vector-based genetic therapies down the road. There had been serious adverse events, even a death, in trials for similar treatments. And yet, the known course of DMD without intervention — disease progression, cardiomyopathy and untimely death — called out to us for action.
In those days of decision-making, my husband and I were just like the couples in “Joy,” on the other side of the desk, told that an experimental treatment may or may not help our child — who himself was born from a previous breakthrough. The difference was, we were making this monumental decision on behalf of a living child who couldn’t possibly grasp the full degree of its ramifications. That made the calculus more complex, but the root of the equation was the same one faced by anyone whose condition warrants a clinical trial: We had a serious problem with a possible solution. Saying yes to science affirmed the possibility of progress over the certainty of our son’s decline. And so, just like the couples in “Joy,” we answered yes, not only for the chance of success for our own child, but for the sake of science and progress for other, future patients.
That humanity behind science is the real joy of this new movie, reminding us that every breakthrough involves countless mothers and fathers — real, live human subjects who make courageous choices to unleash possibility for themselves and others. At a time when fundamental questions are being posed about medical science, let us not forget the beauty of it, the existential poetry and shared humanity that drives us toward the next breakthrough. Let’s remember that no breakthrough is a foregone conclusion; each requires a discovery, then a passing of the torch from the dedicated scientist at the bench to the brave patient in the clinic willing to try it first. As Steptoe, Edwards, Purdy and baby Louise remind us, science is messy. It is hard. It is our shared way forward. It is worth it.
Follow Cognoscenti on Facebook and Instagram. And sign up for our weekly newsletter.

