Support WBUR
Coronavirus Coverage
How One Mass. COVID-19 Clinic 'Got To Yes' On (Most) Staff Vaccinations
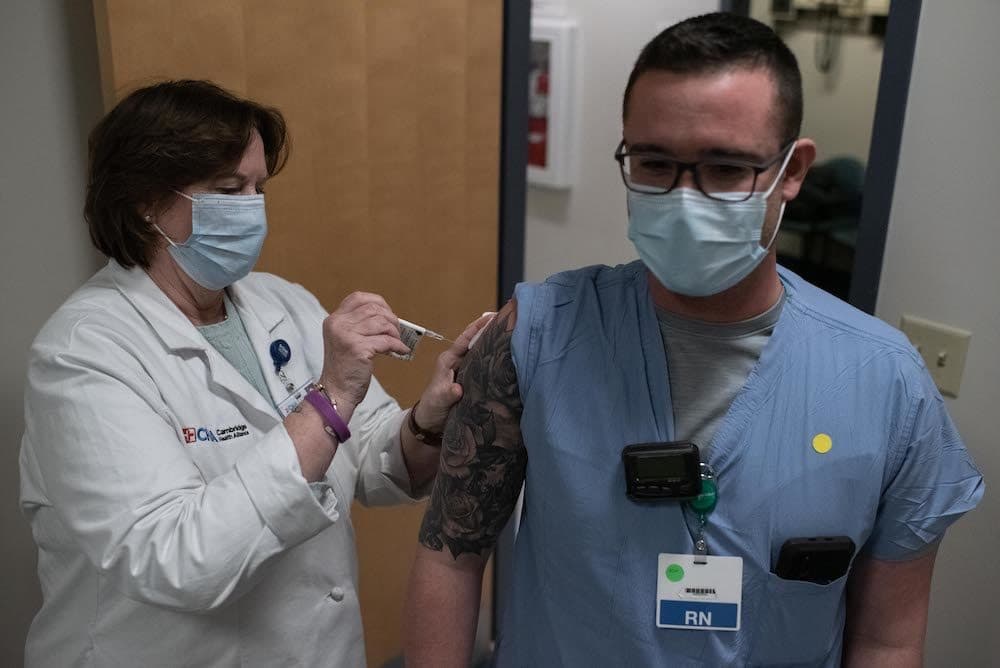
Last month, primary care doctor John Adams realized that coronavirus vaccines would be coming very soon, and he wasn’t ready for them. He didn’t know enough yet to counsel patients or make his own decision about whether to get one. And some of his colleagues at the Cambridge Health Alliance COVID-19 clinic in Somerville, Mass., felt the same.
"It was very much like, ‘We need to learn more about how these things work, because they’re coming fast,' " Dr. Adams said, "and we all sort of shared these doubts."
They also shared unusually intense experience with COVID-19: The clinic has treated thousands upon thousands of patients since March, and its staff knows better than most how devastating the virus can be.
So Adams and physician assistant Neha Sandeep did some research. And at a meeting earlier this month, they presented their findings to the clinic’s medical staff — including their conclusion that although it took just one year instead of the usual 10 or 15 to develop the first coronavirus vaccines authorized for use, that didn’t mean corners had been cut.
"So these vaccines have been developed under a sort of a rapidly accelerated timeline," Adams explained to colleagues. "So — if you go to the next slide — the way that that’s happened is basically by doing everything super fast and also sort of stacking these various phases."
The presentation also addressed the newness of the mRNA technology the first two vaccines use: "Before going into this presentation, I was kind of worried about the idea [that with] these mRNA vaccines, you’re injecting genetic material into the cells," he said. "I was worried: Could this cause cancer?"
"But actually safety is a big advantage of the mRNA vaccines," Adams explained, because they trigger the immune system without the risk of using an actual virus to do it, and they have been tested for years in humans — though against cancer rather than viruses.
By the end of the 45-minute presentation, one staff member told Adams he was a good salesman — which hadn’t been his goal at all, he said.
"Personally, I wasn’t a true believer before doing the research [for] the presentation," he said. "It was only afterwards that I was quite impressed with how these vaccines seem to work."
And so were his medical colleagues, when he laid out the data.
"It was like an 'aha!' moment for all of us, I think," said Dr. Gerard Coste, another primary care physician at the clinic. He and others did have some fears about the speed of the vaccine approvals and how politicized the process had become, Coste said, but the presentation allayed them.
A few days later, Dr. Coste gave a simplified version of the talk to the non-medical clinic staff that includes secretaries and custodians.
"My presentation was trying to be very fair and say, 'Listen, I started this from a position of 'I don’t know. I want to see the data. I want to see it,' " he said. "And I think that presenting the data and not telling anybody what they should do makes a difference."
Coste also described an atmosphere of widespread trust among a staff that has worked shoulder to shoulder in the coronavirus trenches for months.
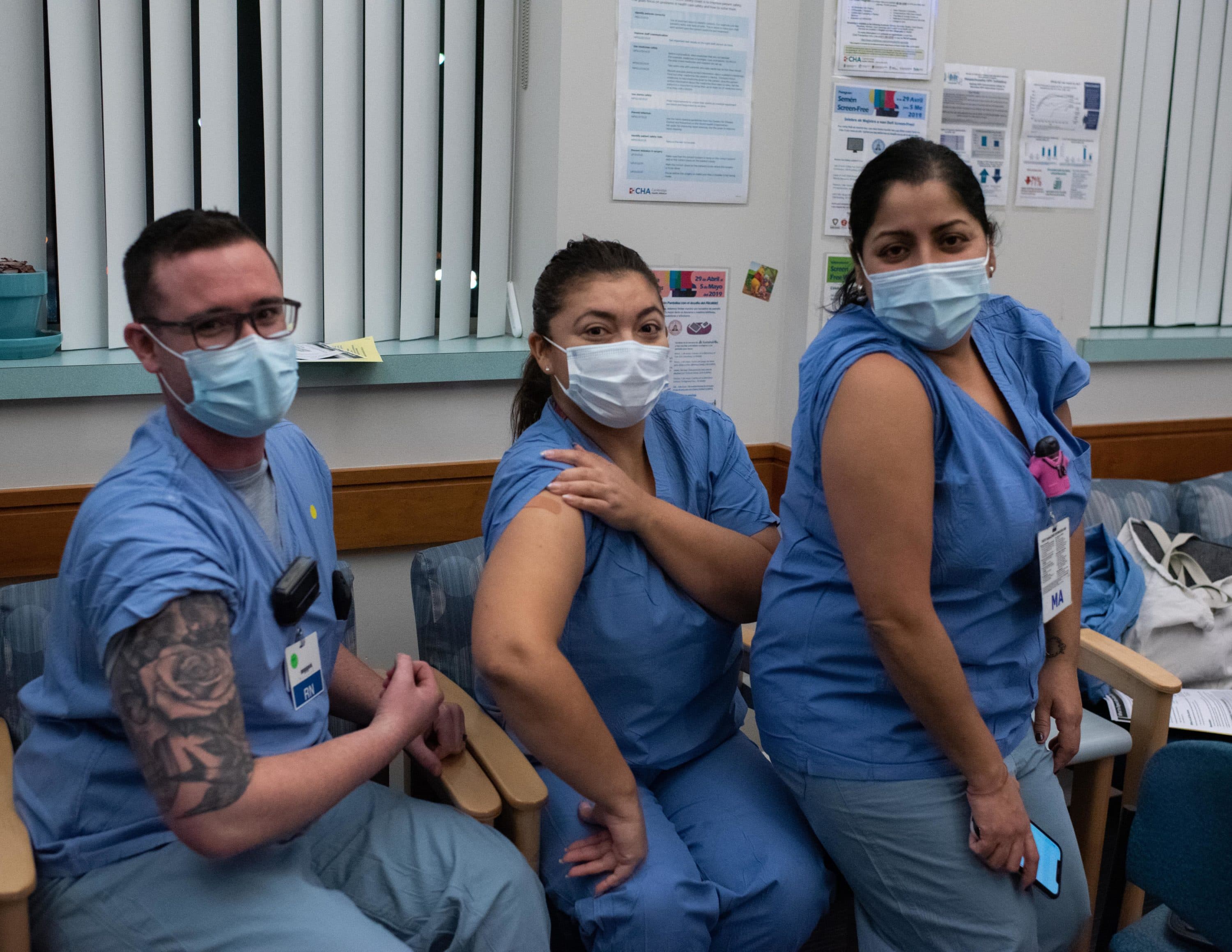
Whatever the factors, when the vaccines arrived at the clinic last week, there were a whole lot of “willing arms.” One doctor, Pieter Cohen, even snowshoed four miles through the nor'easter snow to get to the clinic for his shot.
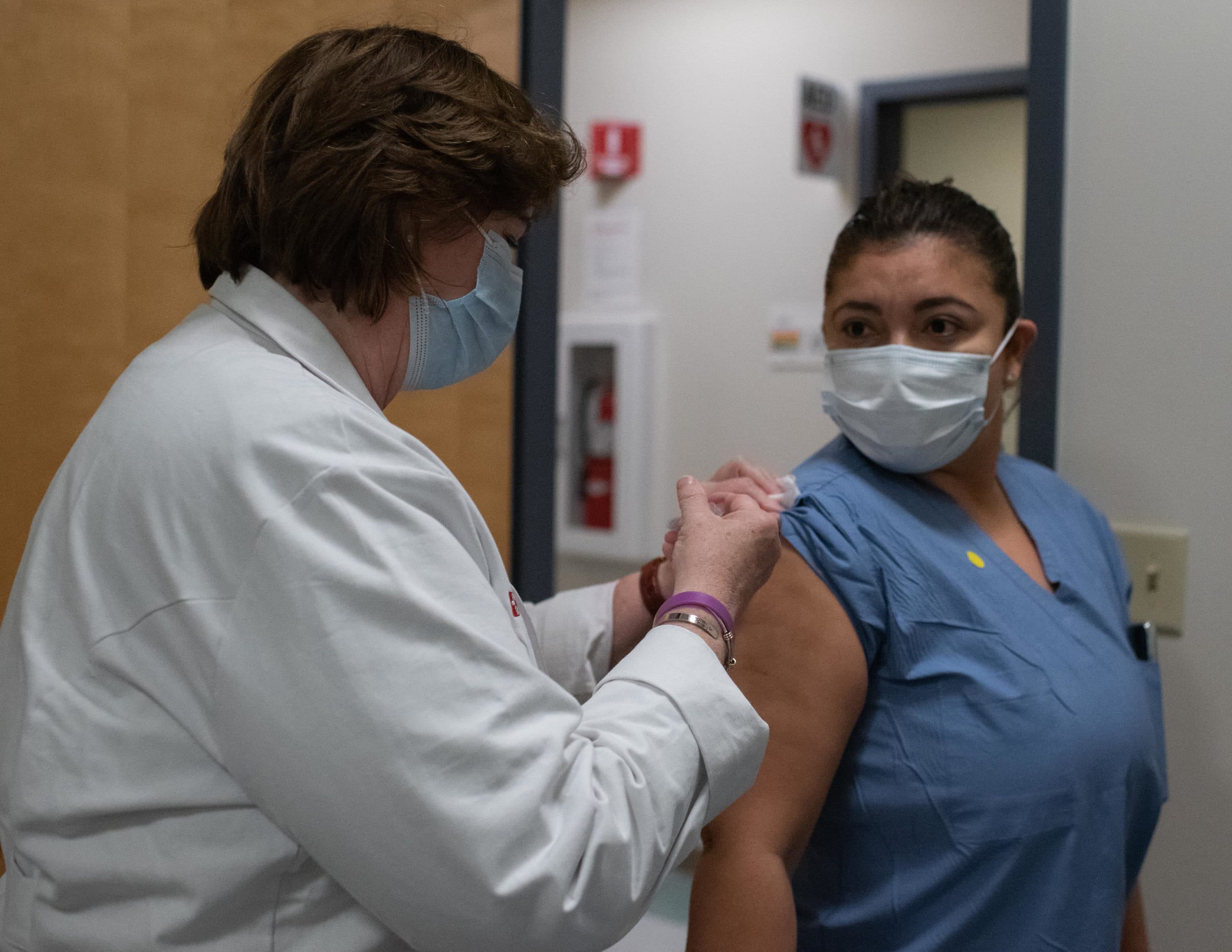
Medical assistant Nancy Morales said at first, she was not eager to get vaccinated because she was scared, and talking to her family didn't help.
"But the fact that I wanted to do it, and I was around people and getting advice from all the docs here," she said, "I said, 'yes, I’m going to just do it.' "
Operations supervisor Elisangela Monteiro said she was initially hesitant, too, "but after seeing Gerard’s presentation last week, I made up my mind."
And physician assistant Neha Sandeep said her own research for the presentation persuaded her.
"Like a 100,000 people have gotten this vaccine in trials and are doing well, and have measurable immunity," she said. "That is incredibly reassuring to me."
All three got the vaccine along with the great majority of the clinic’s 50 or so staffers — but a handful declined. Medical director Janice John said some had concerns about their allergies and the risk of a rare but severe reaction to the injection; but environmental services staff who said no seemed mainly concerned about potential side effects.
"Body aches and muscle aches and discomfort that would make it harder for them to do their work," she said. "But I will also just kind of name this: that there are so many narratives out there about the vaccine."
Narratives that are rife with misinformation about the safety of the vaccines, that is. John added that maybe one lesson from the clinic is that for some people, even data from trusted doctors won’t convince them to get vaccinated. They may need to hear from trusted leaders of their own communities as well.
Meanwhile, she said, if any clinic workers who declined the vaccine change their minds, they’ll still be at the front of the vaccine line.

This segment aired on December 30, 2020.

