Support WBUR
What One 'Extremely Unusual' COVID-19 Case Tells Us About New Variants
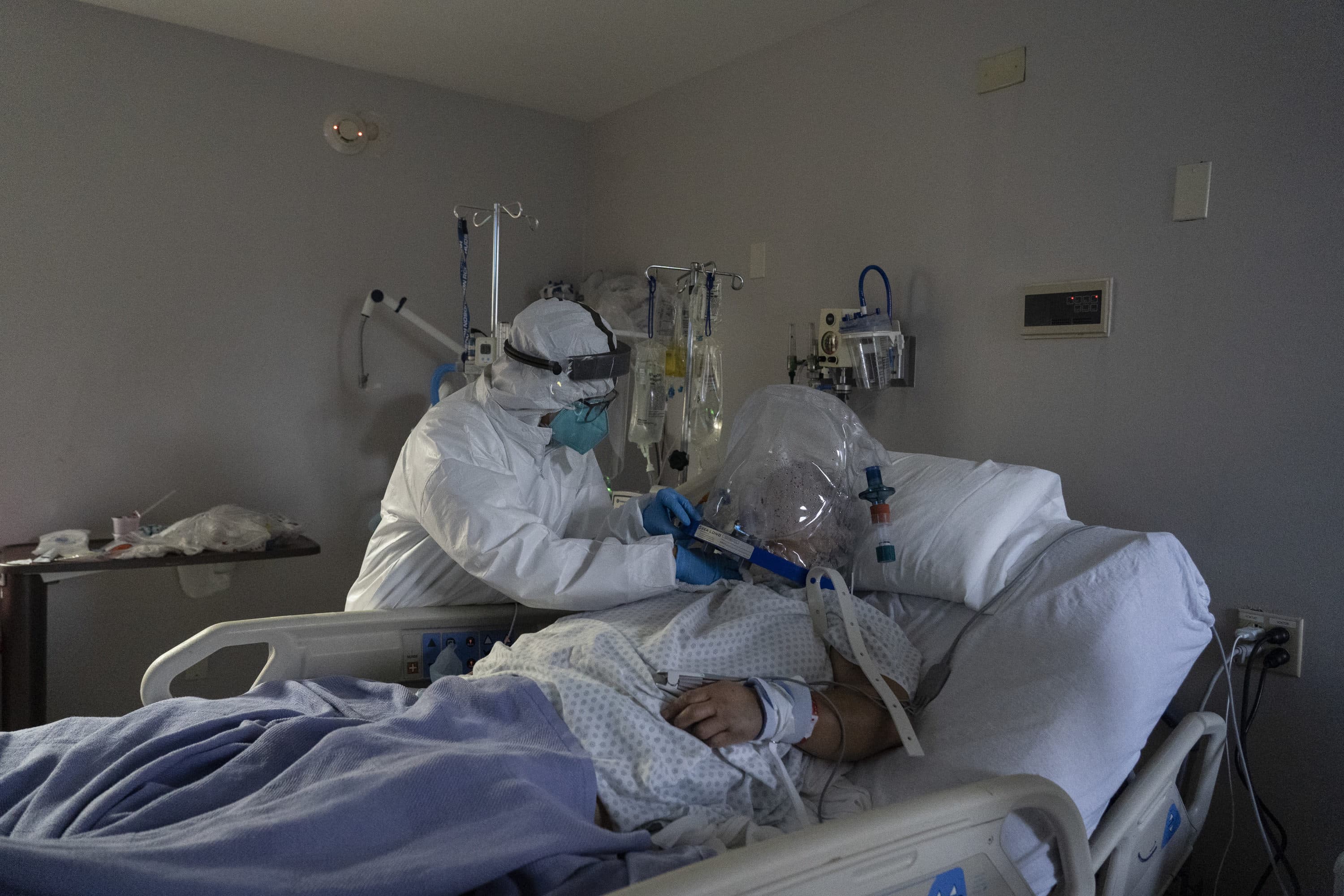
One atypical COVID-19 case is teaching health experts about how the disease is mutating.
A 45-year-old patient in Boston battled COVID-19 for 154 days, one of the longest infections on record, before he died over the summer. Dr. Jonathan Li of the city’s Brigham and Women's Hospital treated this patient and says this man's case is helping doctors understand new emerging variants.
Most COVID-19 patients are able to eradicate the virus and move forward with their lives after leaving the hospital. But in this “extremely unusual” case, the patient was hospitalized with symptoms six times within five months and couldn’t fully get rid of the virus, Li says.
The patient had a disease that required him to take medications that suppress his immune system, making him immunosuppressed and unable to eliminate the infection, Li says. Results from Li’s virology lab determined the patient had what’s called persistent COVID-19.
“When we sequenced the virus over a period of several months, we realized very quickly that this person was not becoming infected with new strains,” he says, “but instead that we saw evidence that the one strain that he was initially infected with was evolving over time.”
The mutation occurred in one particular part of the virus, which then latched on to human cells. When SARS-CoV-2, an RNA virus, replicates itself, it makes mistakes, Li says. These errors can either harm the virus or the patient by helping it replicate faster or improve its ability to persevere inside of its host.
Sequencing the virus revealed several interesting mutations, specifically around a region of the genome known as the spike gene, which coats the outside of the virus and interacts with host cells and antibodies, he says. Researchers saw this outer shell of the virus changing, allowing it to escape the patient’s antibodies, he says.
Some of the new coronavirus variants popping up around the world have an unusual amount of changes in the genome — which reminds Li of this patient’s distinct case.
“The sudden appearance of these variants in different parts of the world really suggests an evolutionary jump from a hidden source of evolution in the community,” he says. “And we don't know exactly how these variants arose in the community, but I think that it is quite possible that the source of some of these variants may have been patients who were immunosuppressed.”
Research efforts haven’t focused on immunosuppressed individuals yet, but Li believes there are more similar cases out there. The findings from this case have many implications for research on public health and infection control measures, he says.
The Centers for Disease Control and Prevention guidelines use symptoms to determine how long a patient should stay isolated, for example, but these recommendations aren’t based on data from immunosuppressed people. Based on Li’s patient and several other published cases, “immunosuppressed patients can have more prolonged viral shedding and more persistent symptoms than an average patient,” he says.
“We should recognize that it's not a one size fits all when it comes to infection control and that we should be more careful about monitoring our patients with immunosuppression,” he says.
Li argues immunosuppressant patients should be a priority when it comes to vaccinations in hopes of slowing down new mutations from forming and spreading.
Many of the new variants are showing changes in the spike gene, which also interacts with the antibodies introduced by vaccines, he says. Studies on some of the vaccines show the overall efficacy drops against some of the new variants.
“It really, I think, speaks to how important it is for us to ramp up vaccine uptake and distribution, not only in the United States but also around the world,” he says, “because if you have uncontrolled infection anywhere in the world, it is a threat to our vaccines and vaccine efficacy everywhere.”
Christina Kim produced and edited this interview for broadcast with Todd Mundt. Allison Hagan adapted it for the web.
This segment aired on February 9, 2021.

