Support WBUR
Coronavirus Coverage
New Trial Suggests Repurposing An Old Drug Might Help Save Lives From COVID-19
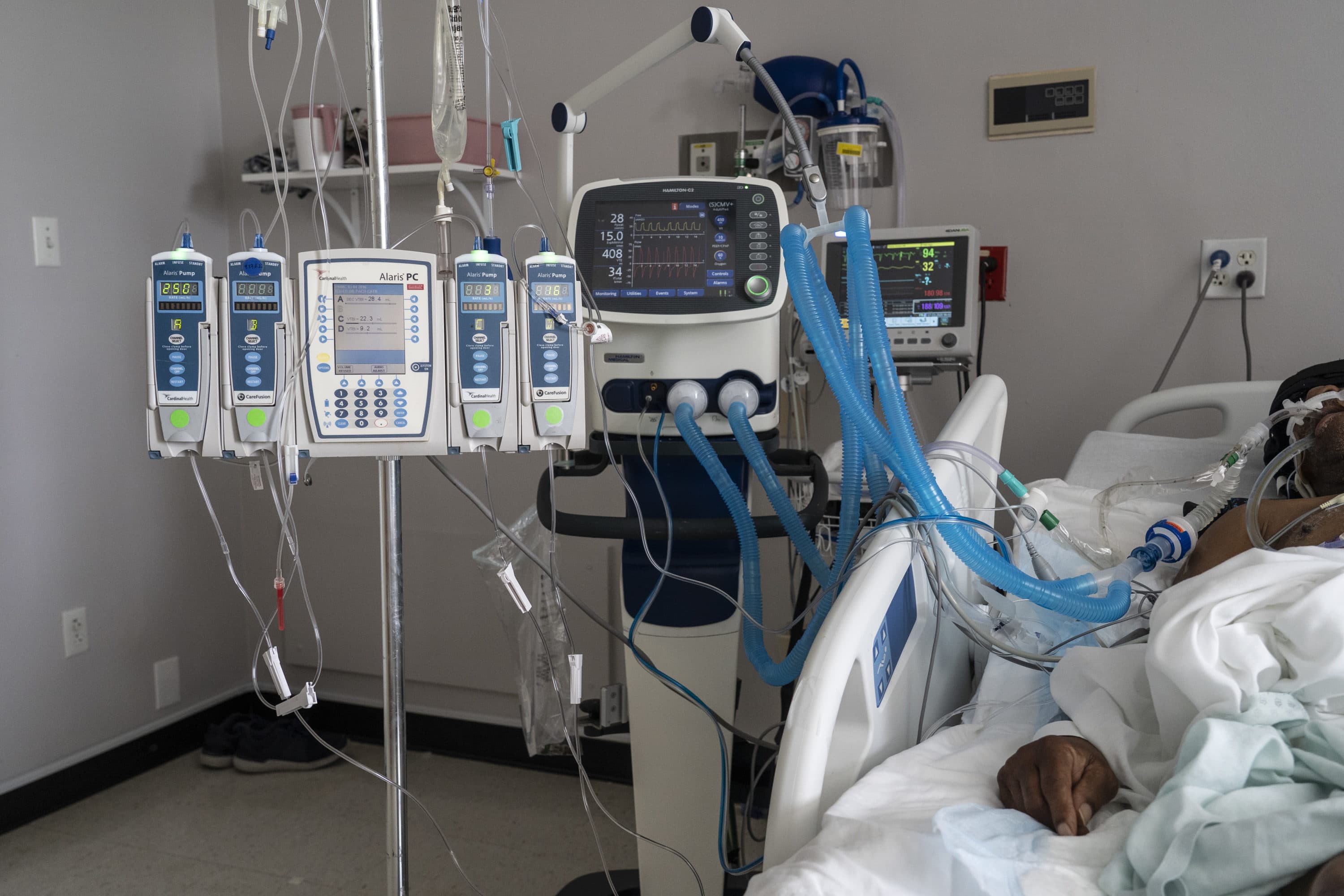
Physicians have learned a lot about COVID-19 in the last year and a half, but that doesn’t mean treating it has gotten much easier. There are only a few drugs proven effective at improving the symptoms of COVID, including the antiviral medication remdesivir and the steroid dexamethasone.
But a drug typically used to boost the immune systems of patients with cancer and bone marrow transplants might be a boon for treating COVID-19, as well as other respiratory infections.
The drug, sargramostim, developed by Lexington-based pharmaceutical company Partner Therapeutics, is a synthetic protein that boosts the body’s production of certain immune defense cells, including some white blood cells. Doctors usually prescribe it for patients who need help rebuilding their immune systems after treatments like chemotherapy, radiation treatment or a bone marrow transplant.
Because COVID-19 can annihilate immune cells in the lungs, Dr. Debasish Roychowdhury, Partner Therapeutics’ chief medical officer, realized the drug might help COVID patients fight off the virus.
So, Roychowdhury reached out to a colleague, University of Utah pulmonologist Dr. Robert Paine, to test sargramostim in a clinical trial with 122 patients severely ill with COVID. Half got the best available treatment, and the other half got the same treatment plus sargramostim. The researchers found that those who received sargramostim had improved oxygen levels. In fact, the study found their levels were roughly three times better than patients who received only the standard treatment.
WBUR spoke with Roychowdhury and Paine about their work, and why they believe sargramostim might be useful not just for COVID-19, but for similar respiratory infections in the future.
This interview has been edited for length and clarity.
How does sargramostim work to help the body fight COVID-19?
Paine: "Sargramostim is a version of a naturally occurring protein that’s in the lung. It’s responsible for maintaining the lung’s defenses all the time because it works on the cells that live in the lung to protect us from infection. Those cells are decimated in the setting of COVID-19. What we’re doing is giving that naturally occurring substance back to the lung in excess amounts to promote those local defense cells.
"We’re giving it by inhalation into the lung, so it can act at the site of this great destruction and help heal the lung and improve oxygen transfer into the body. We found that the COVID patients who got sargramostim showed a very impressive improvement in their respiratory function over six days. We were very excited about this. We think it could help prevent [the use of ventilators for some patients] and the severe respiratory failure that can lead to death in COVID."
Can sargramostim work against the different variants of COVID?
Paine: "There’s been lots of efforts to find antivirals, specific antibodies that might treat COVID. But we found that harnessing the body’s normal defense mechanisms by giving this sargramostim allowed us to show this impressive and exciting improvement in oxygen, and suggests that this is something that can work in the face of new variants that might be resistant to antivirals. It gives us really a greatly improved toolbox to combat COVID."
Roychowdhury: "The biology of this drug in terms of lung disease and health has been worked out over many years. One of the reasons why we started working on COVID was because there was already quite a bit of data from prior work done in other viral infections of the lung or patients with other lung damage like from flu, etc. What we’ve shown is that if the damage is similar to what you see in COVID-19, there may be a benefit of this drug. That’s one of the reasons why the Department of Defense funded this study. That’s why it’s so exciting, because you can take this and start thinking about it more widely and broadly."
Was there a change in mortality or ventilation?
Paine: "The trial was too small for a statistically significant difference in ventilation or mortality, but we did find that fewer people who received sargramostim needed mechanical ventilation. We think an important thing is to move on and do a larger trial to see that. But preventing the worsening of the disease, improving oxygen uptake efficiency, we think that would prevent the severe respiratory failure that causes death."
Is this treatment safe?
Roychowdhury: "That’s one thing about this drug — it’s already approved [by the Food and Drug Administration]. So, it’s a repurposing of an already approved drug. We know the safety profile of this drug, and it’s safe and well tolerated."
How important is it to have more treatment options? Why not just focus on the vaccine effort?
Paine: "Yes, we have had decreased rates of COVID across the United States thanks to vaccination and changes in behavior. But we still have a large number of people dying every day. It’s much lower than it was, but it’s still a large number. The entire world has an enormous burden of COVID, and there is a great worry about emerging variants. So, it’s important to expand our armamentarium to combat COVID, and to do so in a way that’s not dependent on variants or the level of technology in the community has the potential to be a tremendous worldwide benefit.
"Also the fact that it decreased the requirement for oxygen means it might be of benefit in areas where there’s not enough oxygen to treat people, or places where ICUs are limited. There might be significant benefit from sargramostim there. We think the potential is enormous."
