Support WBUR
Human trials for nasal Alzheimer's vaccine to begin in Boston
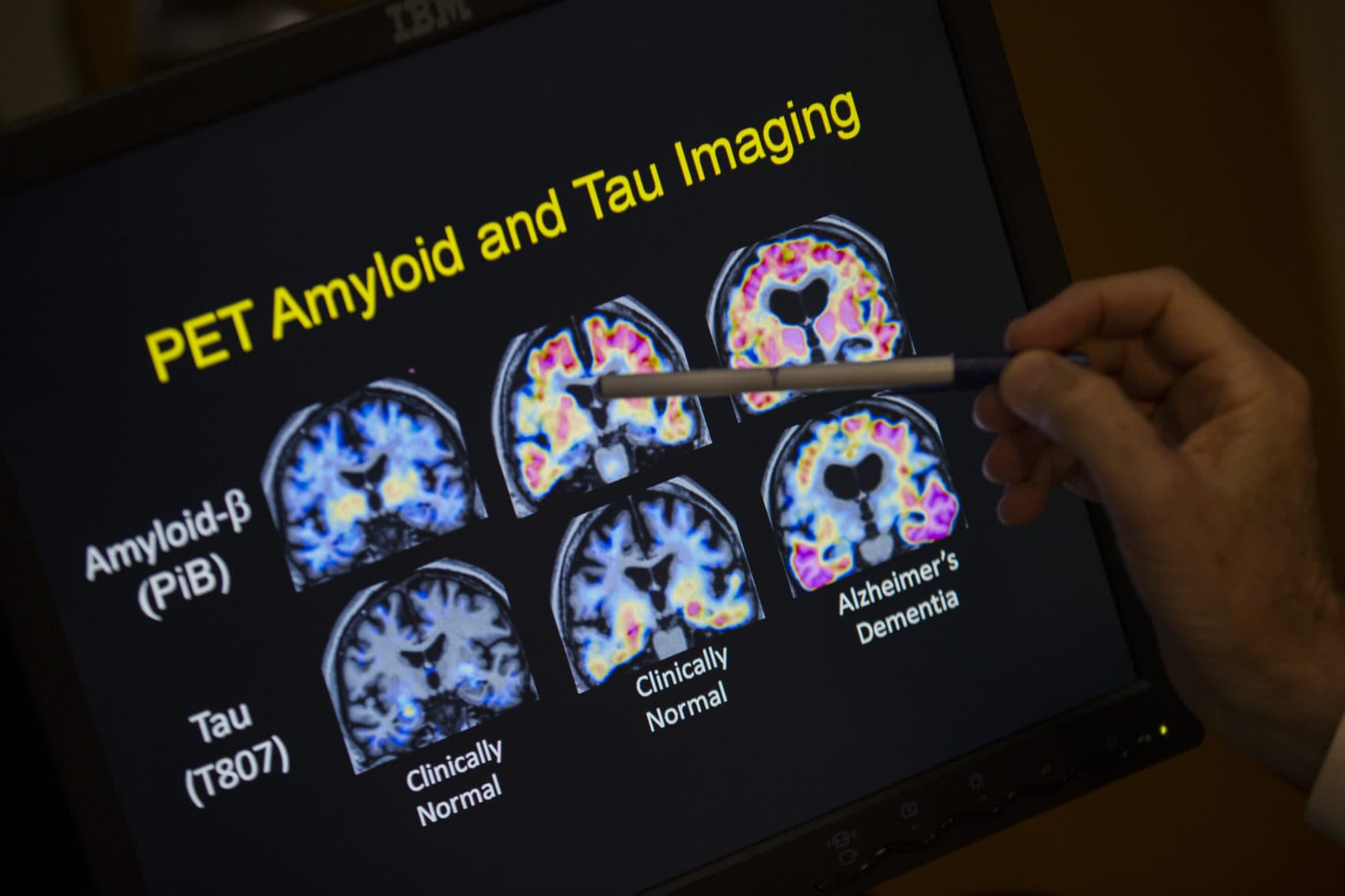
The first clinical trial on people of a nasal vaccine for Alzheimer's disease will begin in Boston next month.
Researchers at Brigham and Women's Hospital say they are now screening patients to select 16 people who will help them evaluate the proper dosage of the vaccine. Later trials could enroll more people and allow researchers to study the vaccine's effectiveness.
The nasal vaccine uses a substance called Protollin, which activates the immune system. Researchers believe it could help the body fight some of the proteins and inflammation believed to contribute to Alzheimer's. Protollin is made up of proteins derived from bacteria and has been used in other vaccines because it helps stimulate certain parts of the immune system.
Dr. Howard Weiner, co-director of the Ann Romney Center for Neurologic Diseases at the Brigham, says trial participants will receive two doses of the vaccine and will then be monitored for a few months.
"We are truly hopeful," Dr. Weiner said. "We've been working on this for a couple of decades and the data we have in animals and initial data testing Alzheimer's patients suggest that there is hope and that this might work."
Weiner says the researchers will eventually study whether the vaccine can help the immune system ward off the development of beta-amyloid plaques, which scientists have identified as a hallmark of Alzheimer's, although there is debate about the role they play in causing the disease. Weiner says the vaccine could potentially help ward off amyloid plaques and other proteins associated with Alzheimer's.
"This could have a broader effect than just amyloid because this would involve the whole immune system," Weiner said.
The trial will involve people between the ages of 60 and 85 who have early symptoms of Alzheimer's. After this trial, Weiner expects a larger clinical trial of patients to study the vaccine's effectiveness in treating, and possibly preventing, Alzheimer's disease.
