Support WBUR
Coronavirus Coverage
Many Hospitals Now Sanitize And Reuse Masks. Workers And Researchers Have Safety Questions
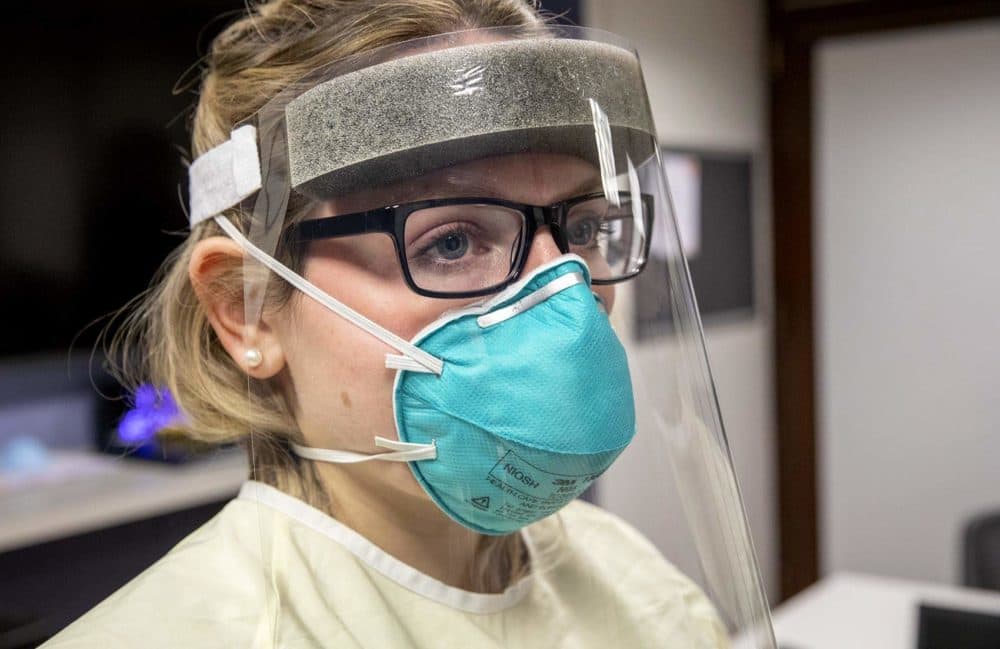
They’ve been stolen, confiscated by federal agents, and flown in secretly from China. N95 masks are in such short supply amid the coronavirus outbreak that hospitals are now sanitizing and reusing face coverings they once tossed in the trash after a single use.
But how many times can a mask be cleaned and reused — and still be safe?
That’s a question nurses, doctors and medical researchers are asking, as hospitals and first responders across the country increasingly turn to decontaminating masks at mass scale, from Boston's most prestigious hospitals to the Los Angeles County sheriff's office. One arm of the federal government is allowing for 20 cycles of cleaning for a single mask. Another suggests three. Many hospitals are touting five to 10.
“One of the concerns we have is that these masks aren’t designed to be reprocessed,” said Richard Peltier, an associate professor at the University of Massachusetts, Amherst, who is working alone in his lab on the deserted campus to find out whether the N95s are degrading each time they are sterilized and reused.
“This is a case where we have to follow the science, and frankly, the science is kind of lacking right now," he added. "I don't necessarily buy that you can reprocess these things 20 times without evidence to support that.”
There’s plenty cause for confusion amid the crush of COVID-19 cases, coupled with political pressure to respond to the scarcity of protective gear available for medical workers.
Partners HealthCare — which runs Massachusetts General Hospital, Brigham and Women’s Hospital and others — has embraced a cleaning system that uses vaporized hydrogen peroxide to decontaminate masks. The initial announcement said masks could be cleaned up to 30 times.
That cleaning system is managed by Battelle Memorial Institute, a multi-billion-dollar Ohio company that operates as a nonprofit and is a major government contractor. Battelle had undertaken a mask-cleaning study back in 2016, but in March pushed hard for approval by the Food and Drug Administration, as coronavirus cases began to surge. Ohio members of Congress and the state’s lieutenant governor urged the agency to approve the system, according to public records and remarks by Ohio Gov. Mike DeWine.
In the wee hours of March 29, the FDA sent a letter approving the mask-cleaning system for emergency use amid the COVID-19 pandemic — because there were not enough news masks. Based on the evidence available, it said, the system “may be effective at preventing exposure” to airborne particulates, and could be used up to 20 times per mask.
The letter also contained this caveat: “No descriptive printed matter relating to the use of the Battelle Decontamination System may represent or suggest that this product is safe or effective for the prevention or treatment of COVID-19.”
But given the need and urgency, the FDA gave Battelle the green light to clean 10,000 masks a day at its headquarters in Columbus, Ohio. The governor was furious at the limitation; Battelle wanted to process 80,000 masks a day, in locations around the country.
“Needless to say, I was quite angry,” DeWine said at a press conference that same day. “I picked up the phone, I called President Trump.”
Within hours, the president tweeted that he hoped the FDA would approve the sterilizing equipment. And by that evening, the regulators had relented. At 10:45 p.m., DeWine tweeted his thanks to Trump and the FDA.
Days later, Somerville was one of the first sites for a Battelle mask-cleaning operation to launch, and by April 13, the company announced it had a $415 million contract from the Defense Department to roll out 60 machines in an array of cities.
Partners executives have said they’ll likely clean masks up to five or 10 times, rather than 20. But prominent nurses’ groups, both local and national, have raised questions about the safety of sanitized masks. And another arm of the federal government, the National Institutes of Health (NIH), came out with a study suggesting three cleanings was safe — but didn’t test them beyond that.
Vincent Munster, a researcher of viruses at NIH’s Rocky Mountain Laboratories in Hamilton, Montana, was a leader of the mask study. He and his team had been working on other coronavirus-related research since January. Then in March, as the virus was exploding, they shifted to a quick three-week N95 mask project, he said.
They tested four different methods of cleaning, and found that vaporized hydrogen peroxide was best. All the methods worked, Munster said in an interview, including one using UV light that some Boston facilities have adopted. In each case, the mask fabric came out clean and sanitized. But the masks do eventually degrade in various ways, including losing their tight fit on the face.
“You need to know what the strain on that mask is. And I personally don't think you should push that,” Munster said. The available studies have not yet taken into account the unusually long hours medical staff are having to wear masks in the current environment — often all day.
“Do you wear a mask only like half an hour? Or have you worn it a complete day and then [decontaminated] it and then wear it again,” Munster said.
Medical workers don't necessarily get their own masks back. They come back from Battelle with a number, indicating how many cleaning cycles they've been through. If there's one thing that may stop hospitals from trying to reuse masks 20 times or more, it's the elastic bands that hold them to the face; everyone seems to agree they break down at that point, or sooner.
Munster declined to comment on why the FDA is allowing 20 cleanings, while his group had only tested for three. Asked how many times he’d be willing to wear a sanitized N95, he said, “I think I would be comfortable wearing that mask for three times” — and only after making sure it still fit properly.
Researchers interviewed by WBUR said the fast turnabout by the FDA is part of a trend developing during the pandemic. The White House has been pressing regulators to allow the use of certain drugs to fight the virus, for instance, before they’ve been fully vetted.
“I think I would be comfortable wearing that mask for three times.”
Researcher Vincent Munster
In a statement, Brittney Manchester, a spokeswoman for the FDA, said, “We worked with Battelle expeditiously, turned around Battelle’s request in a matter of hours, and issued a new authorization on March 29th.” She also said the FDA is “committed to working with our federal government partners and the private sector to find solutions fast, so that we can get essential medical devices to those needing them to protect against COVID-19.”
Now, millions of medical professionals are counting on the safety of mask-sanitizing.
Peltier, the UMass Amherst researcher, is trying to get hold of masks that have been through the Battelle process, to do further safety testing. He said he’s heard from medical providers who are caring for patients with coronavirus — and concerned about reusing masks.
“If there are front-line medical workers using a sterilized face mask that's unsafe,” Peltier said, “I want them to know about it.”
This segment aired on May 4, 2020.
